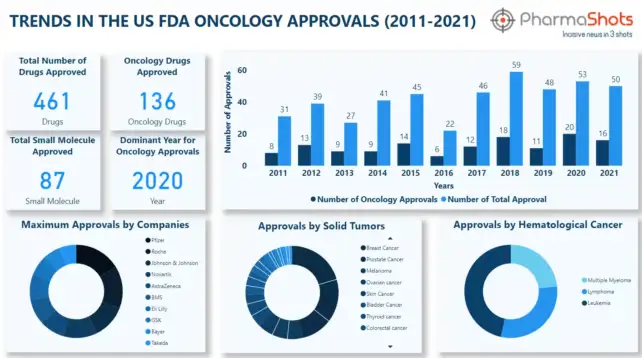
A record number of new oncology drugs have been approved every year, addressing the unique challenges faced by cancer patients. However, oncology is always a thought-provoking area for research and development despite having numerous approved therapies. Many pharmaceuticals are working on oncology and are trying to address the complexity of the disease. We have global…