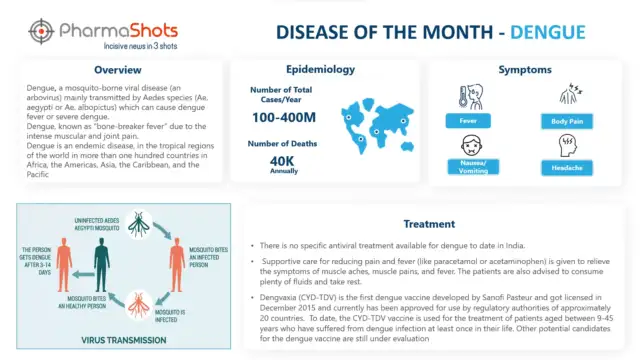
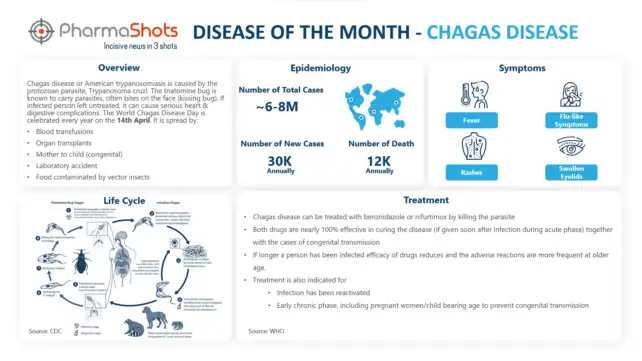
Active Ingredients: Pembrolizumab
Strength: 100 mg/4 mL (25 mg/mL)
Dosage Form: Syringe, Vial
Mechanism of Action: PD-1 ligands antagonists
First Approval: US (Sep 4, 2014), EU (Jul 17, 2015)
Revenue
Keytruda (pembrolizumab) has been a key player in Merck & Co. immuno-oncology drugs for the past years – reaching nearly $17B in annual sales in 2021. Despite the dampening impact…