Getting on with Rare Disease Therapeutic Options (Orphan Disease Therapies)
Shots:
- With the healthcare sector moving ahead with its technological advances, there is still an unexplored section in therapeutics that requires much more attention. Through innovations, life science companies have evolved to make breakthrough discoveries in many focus areas, but when it comes to the rare disease segment, all our endeavors seem to be bleak
- Rare diseases often known as orphan diseases are a matter of concern for decision-makers, HCPs, patient advocacy groups, and most importantly the patients and their families
- PharmaShots brings this month an informative take on rare diseases, the challenges faced in the drug development plans, and available treatment options
With enhanced technological capabilities and pioneering research, life science companies are zestfully exploring the rare disease segment. Rare diseases have distinct definitions across geography. While accrediting the term rare to a particular disease, the prevalence of the disease is examined first. WHO defines a rare disease as one affecting 1 or less, per 1000 population. Usually, chronic genetic diseases were earlier considered to be rare, as they account for about 80% of rare diseases. The group has been broadened to include bacterial or viral infections, allergies, and chromosome disorders. Also known as orphan diseases, the segment accounts for around 7000 types of diseases that affect 300M patients worldwide, of which most patients are young aged. The article focuses on challenges faced in drug development, government incentive programs, grants, and various therapies used to diagnose or treat rare diseases.
How challenging is the drug development plan
Drug development plans for rare diseases can be circuitous, resulting in fewer participation from companies. Some of the major challenges faced by the researchers are:
- Precise Endpoint Selection: Adhering to effective clinical study and endpoint design while reflecting regulatory standards is highly significant in a drug development plan. When it comes to rare diseases, regulatory compliance can sometimes be strict and demanding.
- Patient Availability & Enrolment: Sparse patient population, identification, heterogeneity of disease and the prospects of risks post-treatment affect patient availability in rare diseases. Patient enrolment can be challenging due to perpetual clinical visits that may escalate health problems in patients.
- Patient Records and Historical knowledge: Because these diseases are rare and sometimes don’t get diagnosed at the right time. Clear patient history and segment is unknown, or the information is scarcely available which makes it little difficult to develop a treatment.
- Right Diagnosis: Due to the highly specific nature of the genetic mutation, each inherited rare disease requires a painstaking and inch-perfect diagnostic. A slight variance in the therapeutic may result in serious implications on the health of the patients.
- Data Unavailability: Case studies and medical reports may provide substantial information regarding a disease. But when it comes to regulating the drug impact on patients, pertinent data related to patients seems to be missing.
- Development Cost: Overall drug development cost is similar or sometime more in case of rare and orphan diseases and because of patient size is small. Hence, the cost burden comes on the patients and makes drugs expensive for the patient. 4
Government Incentive Programs
FDA (USA): The orphan drug act was passed in January 1983, with a joint effort of the National Organization for Rare Disorders (NORD), and other organizations. The act includes drugs, vaccines, and therapeutics that are intended to treat a disease affecting <200,000 American citizens. The designation also offers a few benefits:
- Qualified clinical trials get tax credits of up to 50% of R&D costs, R&D grants, waived FDA fees, and protocol assistance, and may also qualify for clinical trial tax incentives
- User fees exemption
- Drug sponsors qualify for seven-year FDA-administered market Orphan Drug Exclusivity (ODE)
Rare Paediatric Disease Priority Review Voucher Program: Started in 2012 under the Food and Drug Administration Safety and Innovation Act (FDASIA), the program specifically targets the special therapies for the treatment of rare paediatric diseases. Under this program, the sponsor gets a priority review voucher for their application
Humanitarian Use Device (HUD) Program: Under section 3052 of the 21st century cures act, any medical device that benefits patients in the treatment or diagnosis of a disease affecting not more than 8000 individuals in the US every year gets an HUD designation for marketing that device.
European Union: In 2000, EU, approved Regulation (EC) No 141/2000 referring to drugs developed to treat rare diseases to as "orphan medicinal products". It received from the EC to get marketing exclusivity in the EU for 10 years. The legislation is administered by the Committee on Orphan Medicinal Products of the European Medicines Agency (EMA).
In late 2007, both FDA and EMA came to a mutual understanding of using a common application process with a distinct approval process, making it easier for manufacturers to apply for orphan drug status. 2
Grant Programs to Support Product Development
Paediatric Device Consortia (PDC) Grants Program: This is an FDA-funded consortia that acts as an advisory body and offers support services to innovators of children’s devices. The support services include business, regulatory consulting, and device testing capabilities.
Orphan Products Grants Program: Under this program, clinical investigators who work on the development of safe and effective medical products for patients with rare diseases get assistance in the form of grants.
Rare Neurodegenerative Disease Grant Program: The program was launched by the FDA in response to Accelerating Access to Critical Therapies for ALS. Under this program, the agency allocates grants and assigns contracts to both public and private entities to cover costs of R&D, and interventions that aim to diagnose, mitigate, prevent or treat ALS and other neurodegenerative diseases in adults and children. 2
Therapies for Rare Diseases
Finding cures and therapies for rare diseases has always been challenging for researchers. Treatments for genetically inherited rare diseases include surgery, bone marrow transplantation, antibody therapy, gene therapy, stem cell therapy, and many more.
Small Molecule Drugs: The R&D of small molecule drugs has always been in the mainstream due to its effective nature and easy administration. With AI-aided research, it becomes relatively easier to select the right compound. For example, Trikafta a small molecule drug developed by Vertex Pharmaceuticals for the treatment of cystic fibrosis, was initiated with the development of the CFTR enhancer ivacaftor on in-vitro screening, forth that a diphasic formulation of lumacaftor and ivacaftor under the name Orkambi and finally the small molecule drug Trikafta was developed.
Gene Therapy: Inherited mutations or mutations caused spontaneously can be the root cause of a new rare genetic disease. UniQure’s Glybera is the first approved gene therapy by the EMA in the year 2012 for the treatment of a rare disease lipoprotein lipase deficiency. Forth that, there have been many breakthroughs in rare diseases. Valoctocogene roxaparvovec, developed by BioMarin Pharmaceutical, for the treatment of hemophilia A. The therapy was granted orphan drug status in 2016 and breakthrough therapy designation in 2017.
Stem Cell Therapy: Medically known as Hematopoietic stem cell transplantation (HSCT), the therapy involves the use of human cells to regulate the immune system. Several pathways to stem cell therapy include fetal liver transplantation, umbilical cord blood transplantation, bone marrow transplantation, and others. Umbilical cord blood transplantation has high prospects and has proved to be highly efficient in the treatment of rare diseases. PREVYMI oral and IV fluids from Merck used in the treatment of CMV infections received FDA approval in 2017. HSCT is gradually getting a hold of medical research for the treatment of a wide range of rare diseases.
Antibody Therapy: Muronomab-CD3 is the first therapeutic mAb approved for organ allograft rejection in 1986. Ablynx NV’s Cablivi (capalacizumab), used to treat adult patients with thrombotic thrombocytopenic purpura (aTTP) received FDA approval in 2019. Alexion Pharm’s Soliris, is a mAB against the terminal complement protein C5 for the treatment of paroxysmal nocturnal hemoglobinuria and was approved by the FDA in 2007 for atypical hemolytic uremic syndrome and myasthenia gravis.
Small nucleic acid drugs: Small nucleic acid drugs have witnessed a spiraled effect in biopharmaceutical companies. Nusinersen, licensed by the FDA is the first approved medication to treat SMA. It works by attaching to the precursor mRNA created by the transcription of the SMN2 gene, changing the RNA splicing pattern, and subsequently enhancing the expression of normal SMN proteins. Milasen, an antisense oligonucleotide that contains 22 nucleotides with the same backbone and glyco-chemical modifications as Nusinersen, can be used to treat Batten disease. About 80% of the antisense oligonucleotide acid drugs are used in the treatment of rare diseases, particularly Duchenne muscular dystrophy (DMD). Eteplirsen, used in the treatment of DMD, is an antisense nucleotide drug and the first FDA-approved gene therapy.
Enzyme Replacement Therapy: ERT was initially created for type 1 Gaucher disease in 1980, which later became a proof-of-principle for the treatment of LSD. Genzyme developed a recombinant form of glucocerebrosidase, which was approved by the FDA in 1994. About 15 enzyme replacement therapy rare illness medications have been approved for international marketing for the treatment of Gaucher disease, Fabray disease, Pompe disease, and mucopolysaccharide storage disease. An enzyme replacement agent, Nexviazyme, was approved by the FDA in 2021 for the treatment of patients with late-onset Pompe disease aged ≥1 year.
Hematopoietic stem cell drugs: Therapeutic drugs prepared from stem cells have revolutionized the treatment of various rare diseases. Moreover, the efficacy of the drugs obtained from the stem cells is relatively high when compared to other methods.
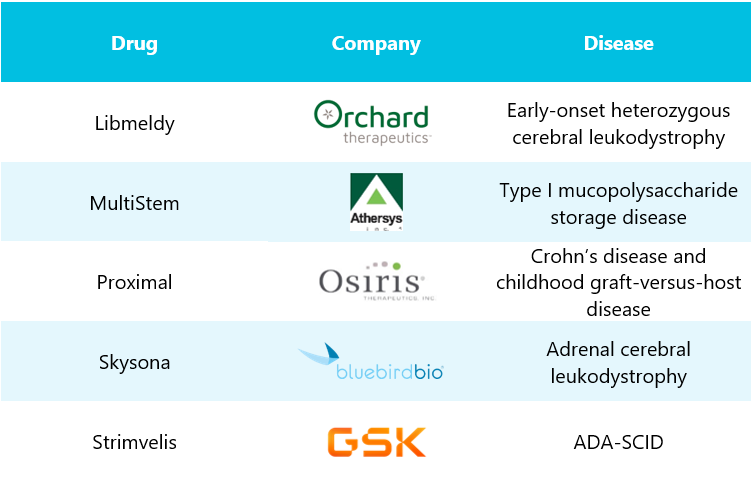
Table 1: Column 1 depicts drugs with company names mentioned in column 2, whereas column 3 represents diseases
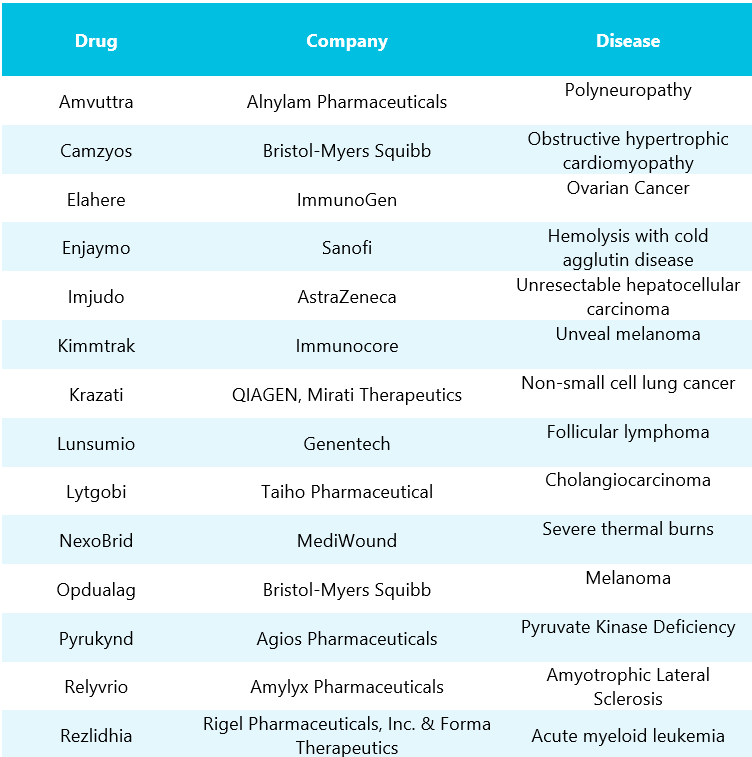
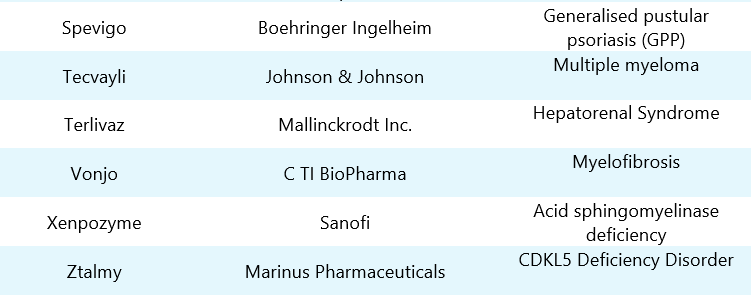
Table 2: List of FDA approved orphan drugs in 2022. Column 1 depicts drugs with company names mentioned in column 2, whereas column 3 represents diseases
Exosome technology-aided therapy
As most gene therapies use AAV as a carrier to deliver curative genetic materials, making patients immune to viruses and leading to ineffective treatment. Since exosome technology does not induce an adaptive immune response, companies are staggering to work with the organizations that offer this technology. One such cooperating endeavor can be seen with the joint initiative of Sarepta Therapeutics (specializes precision medicine and gene therapy) and Codiak (specializes in exosome technology). Exosomes-aided gene therapy brings positive prospects for combined therapy. The extolled combined therapy emanates bright hopes for the treatment of rare diseases, with researchers jointly working on gene technology and stem cell therapy, four hematopoietic stem cell gene therapies have been launched, three of which are used in the treatment of rare diseases. 3
Conclusions & Perspectives
Therapeutics for rare diseases are gaining popularity among the pharmaceutical sector and life science companies. Owing to regulatory relaxations in the form of incentives and grants, giant life science companies are extending their research towards rare diseases. With wide therapies encompassing various aspects of rare diseases, no single therapy can be deemed to be the best. Advancement in each therapy area of rare disease is a step taken to curb it.
References
1. fda.gov
2. Frontiersin
3. National Center for Biotechnology Information
4. Global Forum
Related Post: Molecular Farming: Paving a Modern Track for Pharmaceutical Sectors (Molecular Pharming)
Tags

Saurabh is a Senior Content Writer at PharmaShots. He is a voracious reader and follows the recent trends and innovations of life science companies diligently. His work at PharmaShots involves writing articles, editing content, and proofreading drafts. He has a knack for writing content that covers the Biotech, MedTech, Pharmaceutical, and Healthcare sectors.