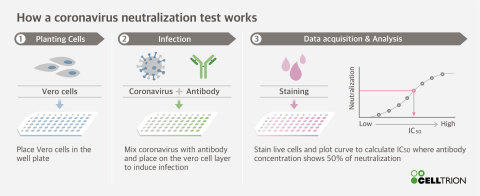
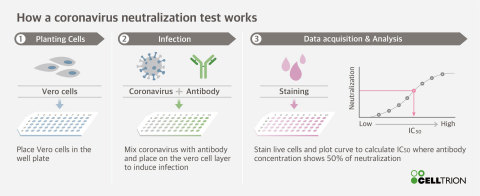
Celltrion Completes Neutralisation Test on Candidate Monoclonal Antibodies (mAbs) for COVID-19 Antiviral Antibody Treatment
- ?Celltrion completes selection of therapeutic antibodies which are most potent for neutralising the virus
- Celltrion expects cell-line development to commence this week
- Celltrion also plans to develop a super-antibody or an antibody cocktail therapy, to target potential mutations of SARS-CoV-2
April 13, 2020 04:00 AM Eastern Daylight Time
INCHEON, Korea--(BUSINESS WIRE)--Celltrion Group today announced that the company has successfully selected the most potent antibody candidates to neutralise SARS-CoV-2, the virus causing COVID-19.
Through a partnership with the Korea Centers for Disease Control and Prevention (KCDC), Celltrion initially identified and secured 300 different types of antibodies that bind to the SARS-CoV-2 antigen. These were then screened based on their ability to bind to the virus spike protein. Celltrion was then able to capture a total of 38 potent neutralising antibodies, of which, 14 are powerful neutralising antibodies against SARS-CoV-2.
Following the selection of antibody candidates which demonstrate high potency in neutralising SARS-CoV-2, Celltrion will begin cell-line development. Once this is completed, Celltrion aims to roll out mass production of the therapeutic antibody and, together with the KCDC, Celltrion will conduct efficacy and toxicity testing in mice and non-human primates.
Ki-Sung Kwon, Head of R&D Unit at Celltrion said, ?We are bringing our full resources and expertise to overcome this global health crisis and are glad to have identified these antibodies sooner than previously expected. These antibodies can recognise multiple epitopes, thus increasing the probability of nonspecific antigen binding. Given the expedited development process of our antiviral antibody treatment, we anticipate moving to first-in-human clinical trials in July. We are also on track with the development of a ?super antibody? or ?an antibody cocktail? and the launch of a rapid self-testing diagnostic kit in the summer of this year.?
Notes to Editors:
About Celltrion Healthcare
Celltrion Healthcare is committed to delivering innovative and affordable medications to promote patients? access to advanced therapies. Its products are manufactured at state-of-the-art mammalian cell culture facilities, designed and built to comply with the US FDA cGMP and the EU GMP guidelines. Celltrion Healthcare endeavours to offer high-quality cost-effective solutions through an extensive global network that spans more than 110 different countries. For more information please visit:?https://www.celltrionhealthcare.com/en-us
About COVID-191
Coronaviruses (CoV) are a family of viruses that lead to illnesses from the common cold to severe diseases such as Middle East Respiratory Syndrome (MERS-CoV) and Severe Acute Respiratory Syndrome (SARS-CoV). Novel coronavirus SARS-CoV-2 is responsible for the disease COVID-19, this new strain, discovered in 2019, is behind the ongoing pandemic outbreak.
Common signs of COVID-19 include respiratory symptoms, fever, cough, shortness of breath and breathing difficulties. In severe cases, infection can lead to pneumonia, severe acute respiratory syndrome, kidney failure and ultimately death.
Please find up to date information about the outbreak via the World Health Organization at?https://www.who.int/emergencies/diseases/novel-coronavirus-2019.
FORWARD LOOKING STATEMENT
Certain information set forth in this press release contains statements related to our future business and financial performance and future events or developments involving Celltrion/Celltrion Healthcare that may constitute forward-looking statements, under pertinent securities laws.
These statements may be identified by words such as ?prepares?, ?hopes to?, ?upcoming?, ?plans to?, ?aims to?, ?to be launched?, ?is preparing, ?once gained?, ?could?, ?with the aim of?, ?may?, ?once identified?, ?will?, ?working towards?, ?is due?, ?become available?, ?has potential to?, the negative of these words or such other variations thereon or comparable terminology.
In addition, our representatives may make oral forward-looking statements. Such statements are based on the current expectations and certain assumptions of Celltrion/Celltrion Healthcare's management, of which many are beyond its control.
Forward-looking statements are provided to allow potential investors the opportunity to understand management?s beliefs and opinions in respect of the future so that they may use such beliefs and opinions as one factor in evaluating an investment. These statements are not guarantees of future performance and undue reliance should not be placed on them.
Such forward-looking statements necessarily involve known and unknown risks and uncertainties, which may cause actual performance and financial results in future periods to differ materially from any projections of future performance or result expressed or implied by such forward-looking statements.
Although forward-looking statements contained in this presentation are based upon what management of Celltrion/Celltrion Healthcare believes are reasonable assumptions, there can be no assurance that forward-looking statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Celltrion/Celltrion Healthcare undertakes no obligation to update forward-looking statements if circumstances or management?s estimates or opinions should change except as required by applicable securities laws. The reader is cautioned not to place undue reliance on forward-looking statements.
References
1?Coronavirus.?World Health Organization. Available at:?https://www.who.int/health-topics/coronavirus?Last accessed: April 2020
1?Coronavirus.?World Health Organization. Available at:?https://www.who.int/health-topics/coronavirus?Last accessed: April 2020