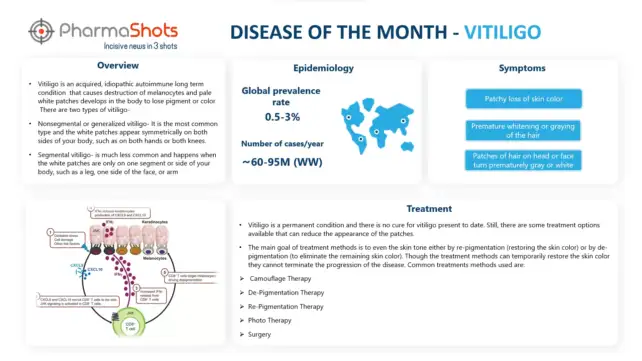
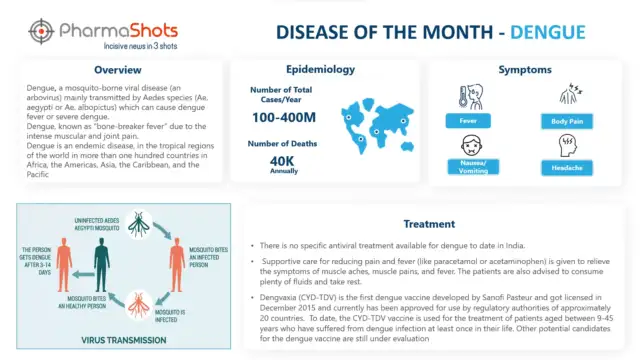
The second quarter of 2022 highlights major acquisitions in the pharma and biotech industry along with multiple approvals. Meanwhile, AstraZeneca’s Farxiga (dapagliflozin) meet its primary endpoints for heart failure with a preserved ejection fraction
Starting with the latest acquisitions, Pfizer acquire Biohaven for ~$11.6B, GSK acquire Affinivax for ~$3.3B. The second quarter of the year…