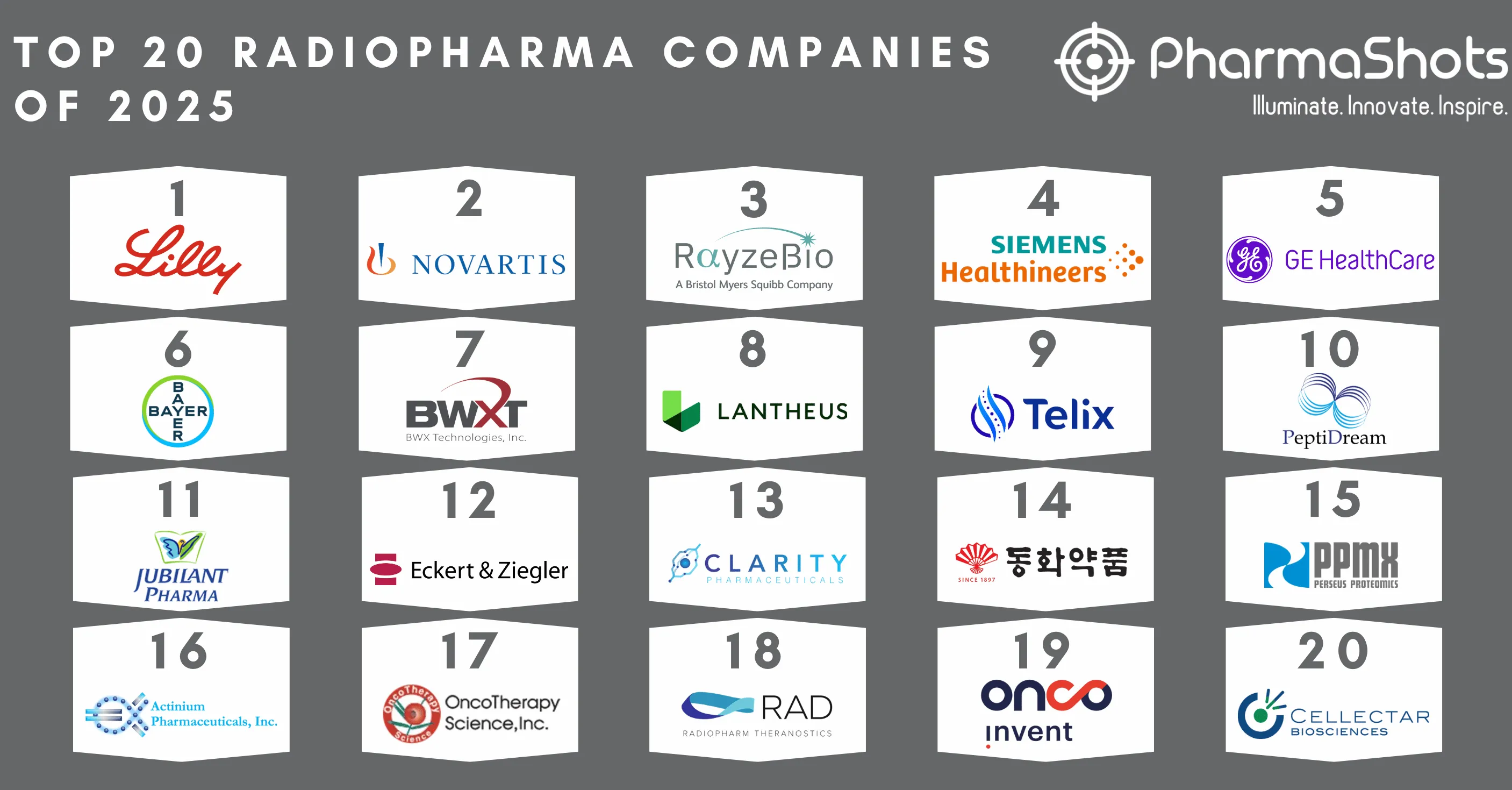
Top 20 Biopharma M&A of 2021 by Total Deal Value
Shots:
- Biopharma merger and acquisition activity continued under the radar with smaller deals through 2021. Despite the absence of eagerly-awaited mega-merger, there were a number of deals above $1 billion.
- CSL Limited holds the top rank by acquiring Vifor Pharma among the top 20 acquisitions with a total deal value of $11.7B. CSL adds 10 commercialized products with a therapeutic focus on areas including Hematology and Thrombosis, Cardiovascular-Metabolic, and Transplant.
- This article is based on 2021 deals data as provided by the DealForma database. Our team at PharmaShots compiled a list of the top 20 acquisitions of 2021 based on total deal value

Deal Date: Nov 11, 2021
Deal Value: $954M
20. Organon & Co. acquired Forendo Pharma. The acquisition strengthens Organon’s women’s health pipeline by adding Forendo’s Phase 2 candidate FOR-6219, an oral HSD17B1 inhibitor for endometriosis and a preclinical program for the polycystic ovarian syndrome (PCOS). Forendo shareholders received $75M up front and are eligible to receive up to $270M in development and regulatory milestones and up to $600M in commercial milestones payments. Organon assumes Forendo’s debt of $9M. In December 2021, Organon & Co. completed the acquisition of Forendo Pharma.
.png)
Deal Date: Jan 4, 2021
Deal Value: $960M
19. Angelini Pharma acquired Arvelle Therapeutics with its lead drug Cenobamate. Arvelle shareholders received $370M up front, an additional $240M following regulatory approval, and are eligible to receive up to $350M upon achieving specific revenue targets for Cenobamate. In February 2021, Angelini Pharma finalized the acquisition of Arvelle Therapeutics.
.png)
Deal Date: July 14, 2021
Deal Value: $1B
18. Lilly acquired Protomer Technologies, including its peptide and protein engineering platform and glucose-sensing insulin program. Protomer shareholders mayreceive >$1B in development and commercial milestones. Lilly currently holds a 14% stake in Protomer Technologies as part of its previous equity investment in Protomer alongside the JDRF T1D Fund.
.png)
Deal Date: Dec 21, 2021
Deal Value: ~$1.225B
17. Sanofi acquired Amunix Pharmaceuticals. Amunix Pharmaceuticals' lead programs were preclinical AMX-818, a masked HER2-directed TCE for cancer, and Pro-XTENTM, XPAT, and XPAC technology used to discover and develop T-cell engagers (TCE) and cytokine therapies for the treatment of cancer. The acquisition supports Sanofi’s efforts in cancer discovery with 20 molecules currently in the pipeline. In addition, Amunix received $1B up front and is eligible to receive up to $225M upon achievement of certain development milestones.
.png)
Deal Date: Jan 11, 2021
Deal Value: ~$1.45B
16. Sanofi acquired Kymab with its fully human monoclonal antibody KY-1005, which can treat immune-mediated diseases and inflammatory disorders. Kymab received $1.1B up front and is eligible to receive up to $350M upon certain milestones. In April 2021, Sanofi completed the acquisition of Kymab.
.png)
Deal Date: Dec 22, 2021
Deal Value: $1.5B
15. Novartis acquired Gyroscope Therapeutics. Gyroscope Therapeutics’ lead programs were Phase II gene therapy GT-005 for geographic atrophy advanced form of AMD and other undisclosed gene therapies. Gyroscope received $800M up front and is eligible to receive up to $700M in additional milestone payments.
.png)
Deal Date: June 2, 2021
Deal Value: $1.7B
14. MorphoSys acquired Constellation Pharmaceuticals. The acquisition strengthens MorphoSys spot in hematology-oncology. In addition, it expands into solid tumors with the addition of Phase-III candidate Pelabresib, a BET inhibitor and CPI-0209, and a second-generation EZH2 inhibitor currently in a Phase II study. Constellation Pharmaceuticals received $1.7B at $34.00 per share at a 1-day premium of 68%. In July 2021, MorphoSys completed the acquisition of Constellation Pharmaceuticals
.png)
Deal Date: Feb 25, 2021
Deal Value: $1.85B
13. Merck acquired Pandion Therapeutics with its precision immune modulators, PT-101 and TALON technology. Pandion Therapeutics received $1.85B at $60.00 per share at a 1-day premium of 134%. In April 2021, Merck completed the acquisition of Pandion Therapeutics
.png)
Deal Date: March 4, 2021
Deal Value: $1.9B
12. Amgen acquired Five Prime Therapeutics. The acquisition gives Amgen Phase 3 ready anti-FGFR2b antibody, bemarituzumab, complements its oncology portfolio, and helps its international expansion strategy, especially in gastric cancer. Five Prime Therapeutics received $1.9B at $38.00 per share at a 1-day premium of 79%. In April 2021, Amgen completed the acquisition of Five Prime Therapeutics.
.png)
Deal Date: Sep 8, 2021
Deal Value: ~$1.9B
11. Sanofi acquired Kadmon Holdings. The acquisition strengthens Sanofi's transplant and rare disease business with the addition of approved rezurock for GVHD. Kadmon shareholders received $1.9B at $9.50 per share at a 1-day premium of 79%. In November 2021, Sanofi completed the acquisition of Kadmon Holdings.
.png)
Deal Date: Aug 5, 2021
Deal Value: $2B
10. Bayer acquired Vividion Therapeutics. The acquisition strengthened Bayer’s small molecule capabilities and granted Bayer full rights to Vividion’s proprietary discovery platform, which comprises synergistic components, chemoproteomic screening technology, an integrated data portal, and a proprietary chemistry library. Vividion shareholders received $1.5B up front and are eligible to receive up to $500M in milestones.
.png)
Date: Aug 23, 2021
Deal Value: $2.26B
9. Pfizer acquired Trillium Therapeutics with its lead molecules TTI-622 and TTI-621 for hematological malignancies. Trillium Therapeutics received $2.22B at $18.50 per share at a 1-day premium of 204%. In November 2021, Pfizer completed the acquisition of Trillium Therapeutics.
.png)
Deal Date: July 27, 2021
Deal Value: $2.5B
8. Amgen acquired Teneobio. The acquisition strengthens Amgen's antibody capabilities and bispecific T-cell platforms with Teneobio's bispecific and multispecific antibody technologies, which compliment Amgen’s BiTE platform. The acquisition also gives Amgen TNB-585, a bispecific T cell-engager that compliments Amgen’s existing prostate cancer candidates acapatamab and AMG 509 and the additional preclinical candidate with the potential to reach IND in the near term. In June 2021, Amgen exercised its option to acquire TeneoOne Inc. Three other Teneobio affiliates (TeneoTwo Inc, TeneoFour Inc and TeneoTen Inc) were spun off to existing equity holders. Teneobio received $900M up front and is eligible for up to $1.6B in contingent milestone payments. In October 2021, Amgen completed the acquisition of Teneobio.
.png)
Deal Date: Feb 1, 2021
Deal Value: $3.05B
7. Horizon Therapeutics acquired Viela Bio for $3.05B at $53.00 per share at a premium of 53%. The acquisition strengthens Horizon’s portfolio of autoimmune and inflammatory diseases by adding Phase III candidate uplizna for myasthenia gravis, Phase II candidates VIB-4920 for kidney transplant rejection and rheumatoid arthritis, and VIB-7734 for autoimmune diseases and a Phase I mAb, VIB-1116. In March 2021, Horizon Therapeutics completed the acquisition of Viela Bio.
.png)
Deal Date: Aug 3, 2021
Deal Value: $3.2B
6. Sanofi acquired Translate Bio for $3.2B at $38.00 per share at a premium of 30%. The acquisition strengthens Sanofi’s spot in vaccine and therapeutics by adding Translate Bio’s mRNA technology platform. In September 2021, Sanofi completed the acquisition of Translate Bio.
.png)
Deal Date: Nov 18, 2021
Deal Value: $3.3B
5. Novo Nordisk acquired Dicerna Pharmaceuticals for $3.3B at $38.25 per share at a premium of 80%. The acquisition gives Novo Nordisk ribonucleic acid interference (RNAi) platform and leads Phase III candidate nedosiran for primary hyperoxaluria and phase II RNAi therapy RG-6346 for chronic hepatitis B virus (HBV) infection and other programs in hepatic and cardiometabolic diseases. In December 2021, Novo Nordisk completed the acquisition of Dicerna Pharmaceuticals.
.png)
Deal Date: Dec 13, 2021
Deal Value: $6.7B
4. Pfizer acquired Arena Pharmaceuticals for $6.7B at $100 per share at a premium of 100%. The acquisition strengthens Pfizer’s portfolio of gastroenterology, dermatology, cardiology, inflammation, and immunology. Arena Pharmaceuticals' lead programs are Phase III etrasimod, a selective sphingosine 1-phosphate (S1P) receptor modulator for the treatment of immuno-inflammatory diseases including gastrointestinal and dermatological diseases, Phase II olorinab, cannabinoid type 2 receptor (CB2) agonist for the treatment of visceral pain, Phase II APD418, a β3-adrenergic receptor (AdrR) antagonist and cardiac myotrope for the treatment of acute heart failure.
.png)
Deal Date: Feb 3, 2021
Deal Value: $7.2B
3. Jazz Pharmaceuticals acquired GW Pharmaceuticals. The acquisition gives Jazz Epidiolex, a product with near-term blockbuster potential developed for seizures associated with Lennox-Gastaut Syndrome (LGS), Dravet Syndrome and Tuberous Sclerosis Complex (TSC). The acquisition also strengthens Jazz’s cancer and neurology portfolio. GW Pharmaceuticals received $7.2B at $220.00 per ADS, where shareholders received $200.00 per share in cash and 0.12036 Jazz ordinary shares or approximately $20.00 at a premium of 50%. Each ADS is equivalent to 12 GW shares. In May 2021, Jazz Pharmaceuticals completed the acquisition of GW Pharmaceuticals.
.png)
Deal Date: Sep 30, 2021
Deal Value: $11.5B
2. Merck acquired Acceleron Pharma for $11.5B at $180.00 per share at a premium of 34%. The acquisition will strengthen Merck’s cardiovascular and hematology portfolio with the addition of Phase III fusion protein Sotatercept for pulmonary arterial hypertension and Reblozyl, an erythroid maturation recombinant fusion protein approved in the US, EU, Canada, and Australia for rare blood disorder developed in partnership with Celgene. In November 2021, Merck completed the acquisition of Acceleron Pharma.
.png)
Deal Date: Dec 14, 2021
Deal Value: $11.7B
1. CSL Limited acquired Vifor Pharma for $11.7B at $179.25 per share at a 1-day premium of 61%. The acquisition expands CSL’s portfolio of renal disease and iron deficiency with the addition of 10 approved products and compliments its hematology and thrombosis, cardiovascular-metabolic, and transplant pipeline. Vifor's lead products were vadadustat, a HIF-PH inhibitor, vamifeport, an oral ferroportin inhibitor for sickle cell disease, Ferinject, for the treatment of iron deficiency and iron-deficiency anaemia, Avacopan, an oral inhibitor of complement C5a receptor (C5aR) for the treatment of ANCA-associated vasculitis.
Related Post: Top 20 Biopharma M&A of 2020 by Total Deal Value
Tags

This content piece was prepared by our former Senior Editor. She had expertise in life science research and was an avid reader. For any query reach out to us at connect@pharmashots.com