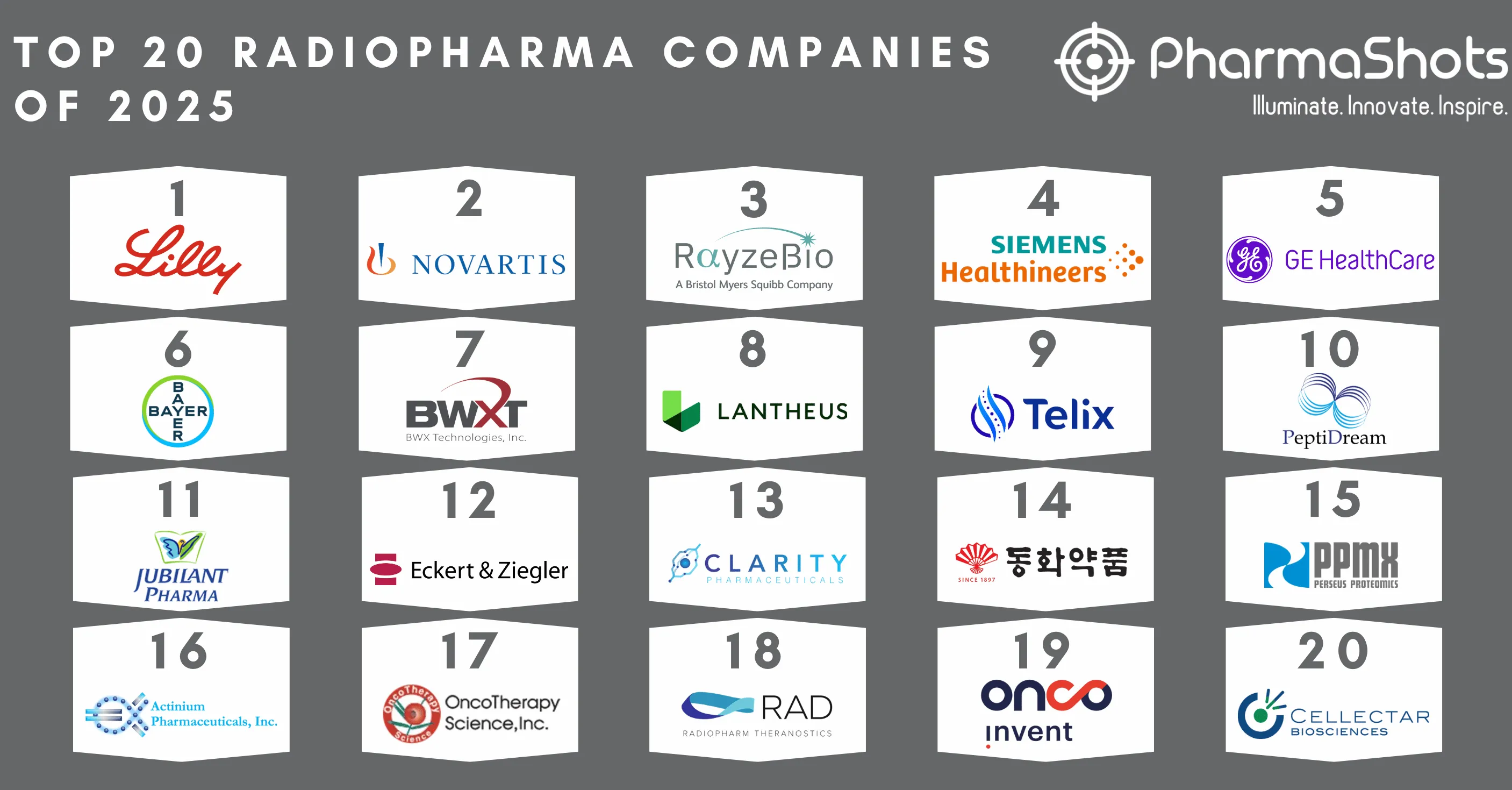
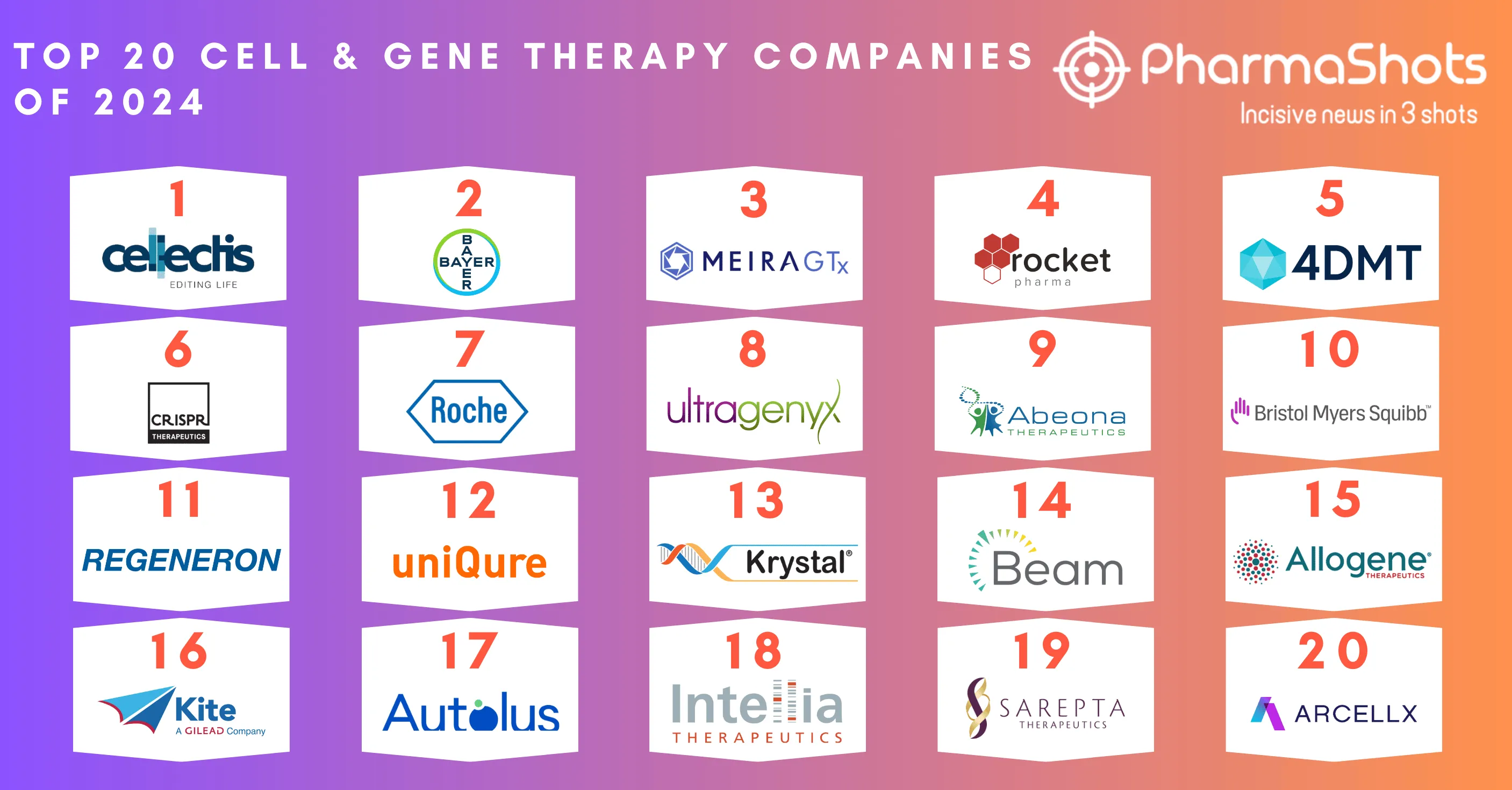
Top 20 Oncology Companies by Total Products of 2019
To remain successful in the oncology market- change is now a key to adapt to this altering market dynamics. A recent development in science and technology platforms are likely to grow faster for cancer treatment in comparison to other therapy areas. However- despite robust levels of pipeline activity- oncology remains a challenging area for research and development. The top oncology companies are now developing more advanced- effective- and tolerable drugs to improve treatment outcomes and patient experience. Our team at PharmaShots has compiled lists of the top 20 oncology companies based on their total product including pipeline and approved products.

Total Oncology Products: 11
Founded Year: 2005
Market Cap: ~$28.08B
Total Employees: ~15-883
Headquarter: Tokyo- Japan
Stock Exchange: TYO
Astellas Pharma is a Japanese multinational pharmaceutical company focused on the therapeutic fields of urology- immunology including transplantation and infectious diseases- oncology- neuroscience and DM complications- and metabolic diseases. Astellas has three approved drugs in its oncology portfolio which include Xtandi- PADCEV & Xospata. It has successfully generated revenue of $3.72B in 2019 from Xtandi for the treatment of prostate cancer. In Dec'2019- the US FDA approved Xtandi for patients with metastatic castration-sensitive prostate cancer (mCSPC).

Total Oncology Products: 11
Founded Year: 1899
Market Cap: ~$62.85B
Total Employees: ~15-000
Headquarter: Tokyo- Japan
Stock Exchange: TYO
Daiichi Sankyo is a Japanese pharmaceutical company that focuses on research and development for developing novel therapies in oncology- including immune-oncology- with further focus on new areas- such as pain management- neurodegenerative diseases- heart- and kidney diseases. The company has 2 approved drugs including Vanflyta and Enhertu. Vanflyta (quizartinib) was launched in Oct'2019 in Japan for the treatment of adult patients with relapsed/refractory FLT3-ITD acute myeloid leukemia (AML). In Dec'2019- Enhertu was approved in the US for HER2 positive unresectable or metastatic breast cancer.

Total Oncology Products: 12
Founded Year: 1891
Market Cap: ~$214.68B
Total Employees: ~71-000
Headquarter: New Jersey- United States
Stock Exchange: NYSE
Merck & Co. is a global health care company delivering innovative health care products with its Prescription medicines- Oncology drugs- Vaccines- Biologic therapies- and Animal Health care products. With 3 approved drugs including Keytruda- Lynparza- and Lenvima- Merck has 12 products in its oncology portfolio. Merck's blockbuster drug- Keytruda used to treat melanoma- lung cancer- head and neck cancer- Hodgkin lymphoma- and stomach cancer has generated revenue of $11.08B in 2019. In Jan'20- FDA approved Merck & Co's Keytruda (pembrolizumab) for patients with BCG-unresponsive high-risk non-muscle-invasive bladder cancer.

Total Oncology Products: 13
Founded Year: 1993
Market Cap: ~$2.78B
Total Employees: ~5-047
Headquarter: Shenyang- China
Stock Exchange: HKD
3SBio is a fully integrated Chinese biotechnology company with market-leading biopharmaceutical franchises in oncology- auto-immune diseases- nephrology- metabolic diseases- and dermatology. There are 3 approved drugs in its oncology portfolio including Cipterbin- Ruisiyi- and Inleusin. In Jun'1995- NMPA production approval documents were obtained for Inleusin- and it was launched in Mar'1996. Cipterbin was formally approved in Jun'2020 as the 1st innovative anti-HER2 monoclonal antibody in China with the engineered Fc region- optimized production process- and a stronger ADCC effect.

Total Oncology Products: 14
Founded Year: 2000
Market Cap: ~$99.90B
Total Employees: ~100-000
Headquarter: Brentford- United Kingdom
Stock Exchange: LON
GlaxoSmithKline (GSK) is a global healthcare company serving the world with drugs- vaccines & consumer healthcare products. With 12 pipeline products and 2 approved products- GSK has 14 cancer drugs in its oncology portfolio including Zejula indicated for the ovarian- fallopian tube- or primary peritoneal cancer and BLENREP used to treat relapsed or refractory multiple myeloma. In Apr'20- FDA approved GSK's Zejula (niraparib) as 1L monotherapy maintenance treatment for women with advanced ovarian cancer regardless of biomarker status.

Total Oncology Products: 14
Founded Year: 1887
Market Cap: ~$394.13B
Total Employees: ~132-100
Headquarter: New Jersey- United States
Stock Exchange: NYSE
Johnson & Johnson (J&J) is an American multinational healthcare company focused on the development and commercialization of pharmaceutical- medical devices- and consumer packaged products. The pharmaceutical portfolio offers products for Cardiovascular- Endocrinology- Immunology- Neuroscience- and Oncology. J&J has 14 drugs in its oncology portfolio with 6 approved products including Darzalex- Imbruvica- Velcade- Zytiga- Balversa- and Niraparib. Imbruvica- an approved small molecule drug used to treat B cell cancers like mantle cell lymphoma- chronic lymphocytic leukaemia- and Waldenstrom's macroglobulinemia and jointly marketed by Janssen Biotech and Pharmacyclics (AbbVie) has generated the revenue $3.41B in 2019. In Apr'19- FDA approved Johnson & Johnson's Balversa (erdafitinib) for advanced or metastatic urothelial carcinoma.

Total Oncology Products: 15
Founded Year: 2010
Market Cap: ~$21.91B
Total Employees: ~4-200
Headquarter: Changping District- China
Stock Exchange: HKG
BeiGene is a global commercial-stage research-based biotechnology company focused on developing small molecules- monoclonal antibodies- molecularly targeted- and immuno-oncology drugs. BeiGene has a portfolio of 15 oncology drugs with 13 pipeline drugs and 2 approved drugs including Brukinsa to treat mantle cell lymphoma and Tislelizumab to treat solid tumours. In Nov'19- FDA granted Accelerated Approval to BeiGene's Brukinsa (zanubrutinib) for patients with mantle cell lymphoma who received at least one prior therapy.

Total Oncology Products: 15
Founded Year: 1901
Market Cap: ~$145.35B
Total Employees: ~33-625
Headquarter: Indiana- United States
Stock Exchange: NYSE
Eli Lilly and Company is a global pharmaceutical firm focused on delivering therapies in two divisions Human Pharmaceutical Products and Animal Health products. The pharmaceutical portfolio offers products for Cardiovascular- Endocrinology- Immunology- Neuroscience- and Oncology. Eli Lilly has 7 approved drugs in its oncology portfolio including Cyramza- Verzenio- Erbitux- Portrazza- Gemzar- and Retevmo. Lilly's Alimta- an approved drug for pleural mesothelioma and non-small cell lung cancer has generated a revenue of $2.11B in 2019. In May'20- Eli Lilly's Retevmo (selpercatinib) received the US FDA's approval to treat advanced RET-driven lung and thyroid cancers.

Total Oncology Products: 17
Founded Year: 1863
Market Cap: ~64.27B
Total Employees: ~103-824
Headquarter: Leverkusen- Germany
Stock Exchange: ETR
Bayer is a leading life science firm with three divisions pharmaceuticals- consumer health- and crop science. Bayer has 11 pipeline drugs and 6 approved drugs in its oncology portfolio including Darolutamide- Copanlisib- Regorafenib- Radium-223 Dichloride- Larotrectinib and Nexavar. With the revenue of $0.79B in 2019- Nexavar used to treat liver cancer- thyroid cancer- or kidney cancer is the highest cancer selling drug for Bayer. In Sep'19- EMA approved Bayer's Vitrakvi (larotrectinib) for NTRK fusion-positive cancers in Europe.

Total Oncology Products: 18
Founded Year: 1991
Market Cap: ~$20.01B
Total Employees: ~1300
Headquarter: Delaware- United States
Stock Exchange: NASDAQ
Incyte Corp is a global biopharmaceutical firm focused on developing therapies in two categories Oncology and Inflammation & Autoimmune. Incyte has 6 approved drugs in its oncology portfolio including Pemazyre- Jakafi- Monjuvi- Iclusig- Olumiant and Capmatinib indicated for multiple tumour types. In Apr'20- FDA approved Incyte's Pemazyre (pemigatinib) as the novel treatment for adults with previously treated- unresectable LA or metastatic cholangiocarcinoma.

Total Oncology Products: 19
Founded Year: 1987
Market Cap: ~$80.88B
Total Employees: ~11-800
Headquarter: California- United States
Stock Exchange: NASDAQ
Gilead Sciences is a research-based biopharmaceutical company with a broad portfolio of drugs. Gilead has 16 pipeline drugs and 3 approved drugs in its oncology segment including Tecartus- Yescarta- and Zydelig. With the revenue of $0.45B in 2019- Yescarta used to treat B-cell non-Hodgkin lymphoma is the highest revenue generated cancer drug for Gilead. In Jul'2020- FDA has granted accelerated approval to Kite's Tecartus used to treat Mantle cell lymphoma (MCL).

Total Oncology Products: 22
Founded Year: 1925
Market Cap: ~$57.12B
Total Employees: ~49-578
Headquarter: Osaka- Japan
Stock Exchange: TYO
Takeda is a global biopharma company focused on Oncology- Gastroenterology (GI)- Neuroscience and Rare Diseases- and Immunology. Takeda has a total of 22 products in its oncology segment with 10 approved drugs including Velcade- Leuprogel- Ninlaro- Adcetris- Iclusig- Alunbrig- Vectibix- Relumina- Cometriq- and Zejula. Takeda has generated revenue of $1.10B from its approved drug- Velcade used to treat multiple myeloma and mantle cell lymphoma.

Total Oncology Products: 23
Founded Year: 2012
Market Cap: ~161.71B
Total Employees: ~30-000
Headquarter: Illinois- United States
Stock Exchange: NYSE
AbbVie is a global- research and development-based biopharmaceutical company focused on developing innovative advanced therapies. The company is focused on developing products in immunology- oncology- virology- and neuroscience- dermatology. AbbVie has 23 products in its oncology segment with 3 approved drugs including Empliciti- Imbruvica- and Venclexta. Imbruvica indicated for B cell cancers like mantle cell lymphoma- chronic lymphocytic leukaemia- and Waldenstrom's macroglobulinemia has reported a revenue of $4.67B in 2019. In May'2019- FDA granted Breakthrough Therapy designation to AbbVie's Venclexta + obinutuzumab (Gazyva) to treat chronic lymphocytic leukaemia or small lymphocytic lymphoma.

Total Oncology Products: 24
Founded Year: 1973
Market Cap: ~$131.10B
Total Employees: ~100-000
Headquarter: Paris- France
Stock Exchange: EPA
Sanofi is a global healthcare leader in vaccines providing healthcare solutions in 170+ countries around the world. Sanofi is ranked third in the global market and first in EU and Latin America. Sanofi has 7 approved products in its oncology segment including Jevtana- Eloxatin- Taxotere- Zaltrap- Sarclisa- Libtayo- and Clolar. Sanofi has generated revenue of $0.57B in 2019 from its approved drug Jevtana used to treat castration-resistant prostate cancer. In Mar'2020- Sanofi received FDA approval for Sarclisa (isatuximab-irfc) used to treat relapsed refractory multiple myeloma.

Total Oncology Products: 24
Founded Year: 1849
Market Cap: ~$201.05B
Total Employees: ~83-000
Headquarter: New York- United States
Stock Exchange: NYSE
Pfizer is a research-based- global biopharmaceutical company having a vast portfolio including Oncology- Vaccine- and other health care products for the prevention & treatment of untreated diseases. With 11 approved drugs including Ibrance- Sutent- Xtandi- Xalkori- Inlyta- Bosulif- Mektovi+Braftovi- Lorbrena- Talzenna- Bavencio- and Daurismo- Pfizer has 24 products in its oncology portfolio indicated for breast- renal cell carcinoma- gastrointestinal stromal tumour- prostate cancer- and several other tumour types. Ibrance- an approved drug for breast cancer has generated revenue of $4.96B in 2019. In Jun'2019- FDA approved Pfizer's Zirabev (bevacizumab- biosimilar) to treat metastatic colorectal cancer- non-small cell lung cancer- glioblastoma- metastatic renal cell carcinoma- and metastatic cervical cancer.

Total Oncology Products: 28
Founded Year: 1980
Market Cap: ~$143.83B
Total Employees: ~23-400
Headquarter: California- United States
Stock Exchange: NASDAQ
Amgen is one of the leading biotechnology companies developing novel therapies focused on cardiology- oncology- neurology- nephrology- and inflammatory diseases. Amgen has a total of 28 drugs with 21 pipeline drugs and 7 approved drugs including Blincyto- Vectibix- Kanjint- Kyprolis- Mvasi- Xgeva- and Imlygic. Amgen's Xgeva used to prevent bone problems in patients with multiple myeloma or from solid tumours has generated sales of $1.93B in 2019.

Total Oncology Products: 39
Founded Year: 1896
Market Cap: ~ $315.6B
Total Employees: ~98-000
Headquarter: Basel- Switzerland
Stock Exchange: SWX
Roche Holding AG is a Swiss multinational healthcare company that operates worldwide under two divisions: Pharmaceuticals and Diagnostics. Roche's oncology portfolio is currently focusing on breast- bladder- cervical- ovarian- liver cancer with 26 pipeline drugs- and 13 approved drugs including Herceptin- Perjeta- Tecentriq- Kadcyla- Alecensa- etc. Roche's Avastin used to treat several cancers and a specific eye disease has generated revenue of $7.30B in 2019. In Aug'2019- FDA approved Roche's Rozlytrek (entrectinib) for ROS1+ NSCLC and granted accelerated approval for solid tumours having NTRK gene fusion.

Total Oncology Products: 62
Founded Year: 1996
Market Cap: ~$219.56B
Total Employees: ~109-000
Headquarter: Basel- Switzerland
Stock Exchange: SWX
Novartis is a multinational group of companies specializing in the research- development- manufacture- and marketing of drugs. Novartis has the highest number of approved drugs in the oncology segment including Tasigna- Sandostatin- Afinitor/Votubia- Promacta/Revolade- Tafinlar + Mekinist- Gleevec/Glivec- Jakavi- Afinitor Disperz/Votubia- Votrient- Kisqali- Lutathera- Kymriah- Piqray- Arzerra- Farydak- Femara- Mekinist- Proleukin- Rydapt- Tabrecta- Tafinlar- Tyverb- Zometa- Zykadia. With a revenue of $1.88B in 2019- Tasigna is the blockbuster drug for Novartis used to treat Philadelphia chromosome-positive chronic myeloid leukaemia (Ph+ CML). In May'19- Novartis has received FDA approval for Piqray (alpelisib) + Fulvestrant to treat breast cancer.

Total Oncology Products: 63
Founded Year: 1887
Total Employees: ~30,000
Headquarters: New York, United State
Market Cap: ~$133.16B
Stock Exchange: NYSE
Bristol-Myers Squibb is an American pharmaceutical company focused on Oncology, Cardiovascular, Immuno-Science, and Fibrosis. BMS has total of 63 drugs in its oncology segment including 14 approved drugs. BMS’ Revlimid used to treat multiple myeloma and myelodysplastic syndromes has generated the highest revenue of $9.37B in 2019. In Dec’19 BMS announced that it has received EC’s approval based on P-III AUGUMENT study assessing Revlimid + rituximab (r2) vs rituximab + PBO in patients with previously treated follicular lymphoma (FL) or marginal zone lymphoma (MZL).
Related Post: Top 20 Innovative Digital Therapeutics Companies of 2019
Tags

This content piece was prepared by our former Senior Editor. She had expertise in life science research and was an avid reader. For any query reach out to us at connect@pharmashots.com