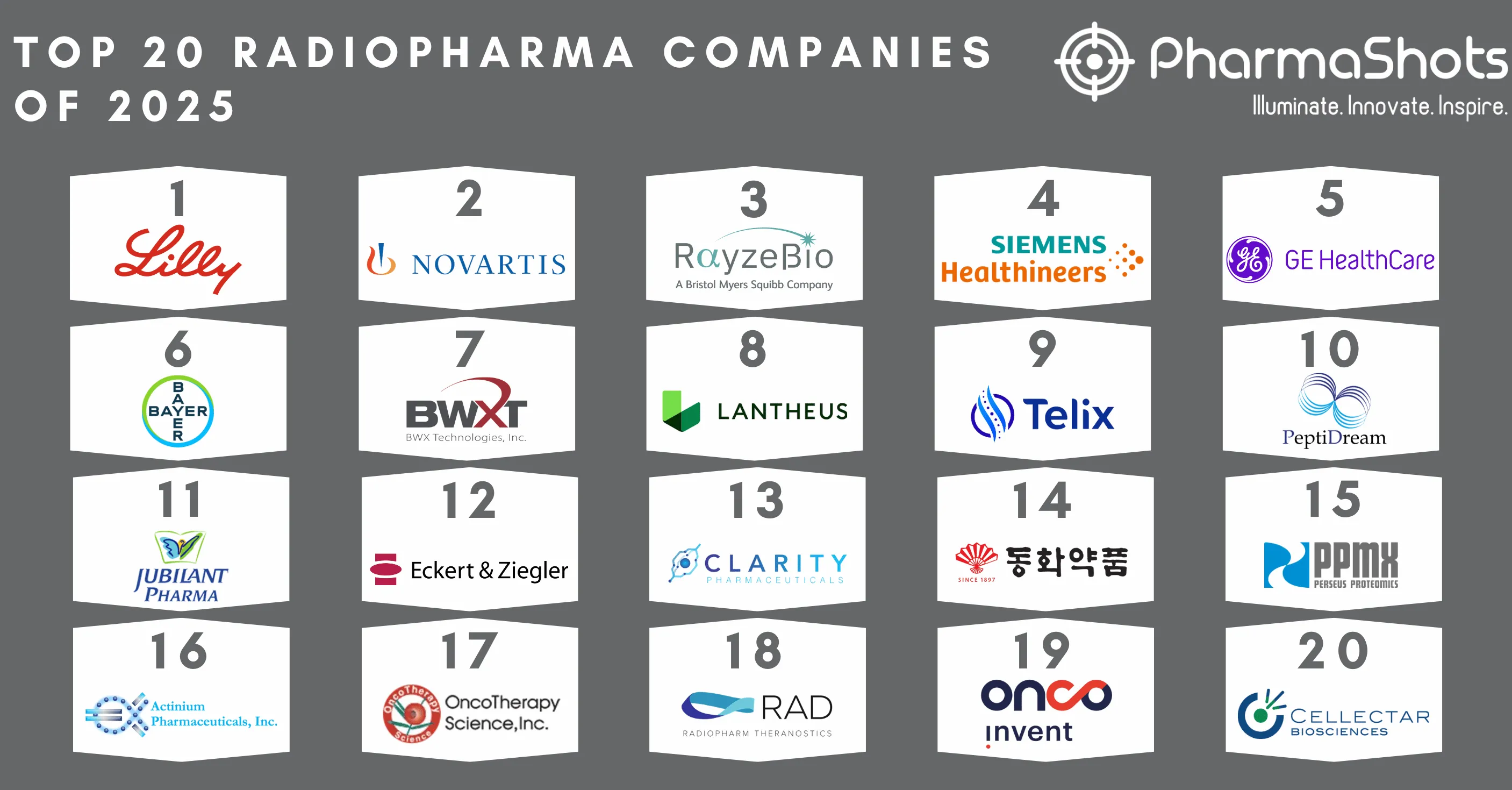
Top 20 Immunology Companies Based on 2020 Immunology Segment Revenue
- Immunology deals with the physiological functioning of the immune system in states of both health and disease as well as malfunctions of the immune system in immunological disorders
- With the new advancement in the immune sector- global pharmaceuticals continue to grow in the field despite the disruption during COVID-19. As in 2019- Abbvie again secured the top position with total revenue of $22.15B- followed by J&J- Roche- and Amgen
- Our team at PharmaShots has compiled a list of the top 20 immunology companies based on their 2020 immunology revenue

Immunology Segment Revenue: $0.015B
Founded Year: 1978
Market Cap: $0.067B
Total Employees: 185
Headquarters: New Jersey- United States
Stock Exchange: NASDAQ
Antares Pharma is a specialty pharmaceutical company focused primarily on the development & commercialization of pharmaceutical products and technologies addressing the unmet needs in the targeted therapeutic areas. The company has reported a total sale of $0.015B from its immunology segment in 2020 from the sales of Otrexup in RA- pJIA- Psoriasis.

Immunology Segment Revenue: $0.09B
Founded Year: 1993
Market Cap: $2.326B
Total Employees: 5,525
Headquarters: Shenyang- China
Stock Exchange: HKG
3S Bio is a Chinese Biotechnology company that focuses on oncology- nephrology- dermatology- and auto-immune diseases. The company unveiled total sales of $0.09B in its immunology segment during the year 2020 from the sales of Yisaipu- which is a TNF inhibitor indicated to treat RA- AS and psoriasis.

Immunology Segment Revenue: $0.99B
Founded Year: 2000
Market Cap: $100.73B
Total Employees: ~99,000
Headquarter: Brentford- United Kingdom
Stock Exchange: LON
GSK is a global healthcare multinational company involved in the development- manufacture- and marketing of pharmaceutical products- vaccines- OTC medicines- and health-related consumer products. With only approved products- Benlysta- GSK has generated a revenue of $0.99B in 2020. In Dec 2020- the US FDA has approved Benlysta as the first medicine for adult patients with active LN.

Immunology Segment Revenue: $1.05B
Founded Year: 2008
Market Cap: $23.61B
Total Employees: 1,600
Headquarters: Dublin- Ireland
Stock Exchange: NASDAQ
Horizon is a biopharmaceutical company that focuses on rare and rheumatic diseases. Horizon has generated the sale of $1.05B from its four approved immunology products including Tepezza- Rayos- Duexis- and Vimovo. During the fiscal year, 2020- Tepezza alone has generated $820M in revenue- followed by Duexis ($125.3M)- Rayos ($71.8M)- and Vimovo ($37.6M).

Immunology Segment Revenue: $1.08B
Founded Year: 2007
Market Cap: $42.9B
Total Employees: 6,805
Headquarters: Osaka- Japan
Stock Exchange: TYO
Mitsubishi Tanabe Pharma Corporation (MTPC) is a Japanese pharmaceutical company that focuses on diabetes- kidney diseases- autoimmune diseases- and vaccines. The company has reported a total sale of $1.08B from its immunology segment in 2020. In Dec'2020 MTPC initiates a long-term OLE trial to the phase III MT-1186-A03 trial of edaravone for Amyotrophic lateral sclerosis

Immunology Segment Revenue: $1.16B
Founded Year: 1891
Market Cap: $190.33B
Total Employees: 58,096
Headquarters: New Jersey- United States
Stock Exchange: NYSE
Merck & Co. is an American multinational pharmaceutical company. The company develops medicines (oncology- OTC- and generic drugs)- vaccines- biologic therapies- and animal health products. In 2020- Merck-approved drugs including Simponi and Remicade generated revenue of $1.16B.

Immunology Segment Revenue: $1.65B
Founded Year: 2005
Market Cap: $30.32B
Total Employees: 15,883
Headquarter: Tokyo- Japan
Stock Exchange: TYO
Astellas is a Japanese multinational pharmaceutical company. It focuses on serving patients' medical needs across multiple therapeutic areas- including cardiology- hematology- immunology- infectious diseases- oncology- and urology. Astellas immune drug- Prograf generated revenue of $1.65B in 2020. In Jan 2020- Astellas announced the construction of a new manufacturing facility for Prograf's API in the Toyama technology center.

Immunology Segment Revenue: $1.93B
Founded Year: 1991
Market Cap: $16.26B
Total Employees: 1,773
Headquarters: Delaware- United States
Stock Exchange: NASDAQ
Incyte is a global biopharmaceutical company that focuses on oncology- inflammation- and autoimmune diseases. In 2020- Incyte has generated revenue of $1.93B in its immunology segment. In Jul 2020- Incyte and MorphoSys announced that the FDA approved Monjuvi (tafasitamab-cxix)- which is indicated in combination with lenalidomide for R/R DLBCL

Immunology Segment Revenue: $1.94B
Founded Year: 1978
Market Cap: $50.50B
Total Employees: 9,100
Headquarter: Massachusetts- United States
Stock Exchange: NASDAQ
Biogen is a US-based biotechnology company that focuses on developing and delivering innovative therapies globally for neurological- neurodegenerative- and autoimmune diseases. Major areas of it include MS- neuroimmunology- neuromuscular disorders including Spinal Muscular Atrophy (SMA)- movement disorders including Parkinson's disease- Ophthalmology- and neuropsychiatry. Biogen generated a revenue of $1.94 with Tysabri in the immunology segment. In Jun'2020- the company submitted the regulatory filing of Tysabri in the US and the EU.

Total Immunology Segment: $2.19B
Founded Year: 1928
Market Cap: $21.81B
Total Employees: 7,600
Headquarters: Brussels- Belgium
Stock Exchange: EBR
UCB is a multinational biopharmaceutical company that specializes in two therapeutic areas- the central nervous system (CNS) and immunology. Cimzia has generated total revenue of $2.1B in 2020. In Jan 2020- UCB's biologic Cimzia (certolizumab pegol) was approved by MHRA for the treatment of PsO- PsA- pustular psoriasis- and psoriatic erythroderma for which existing treatment methods are not sufficiently effective. The approval makes Cimzia the first Fc-free- PEGylated anti-TNF treatment option in Japan.

Immunology Segment Revenue: $2.46B
Founded Year: 1876
Market Cap: $257.75B
Total Employees: 35,000
Headquarters: Indiana- United States
Stock Exchange: NYSE
Eli Lilly is an American pharmaceutical company focused on immunology- neurodegenerative diseases- and endocrinology. Eli Lilly has two approved immunology products including Olumiant and Taltz. Its immune product- Taltz has generated revenue of $1.78B while the remaining is from Olumiant's sales in 2020. Taltz and Olumiant drove the 37% increase in revenue in Immunology.

Immunology Segment Revenue: $3.16B
Founded Year: 1887
Market Cap: $149.73B
Total Employees: 30,250
Headquarters: New York- United States
Stock Exchange: NYSE
BMS is a global biopharmaceutical company that focuses on multiple therapeutic areas including oncology- hematology- cardiovascular- and immunology. Its immune drug- Orencia generated revenue of $3.16 in 2020. The revenue of Orencia increased by 6% and 7% in the US and international markets due to higher demand respectively. In Jun'2020- the company reported results of an open-label P-IV Early AMPLE biomarker study assessing the differences by which Orencia and adalimumab interfere with disease progression in the mod. to sev. RA patients.

Immunology Segment Revenue: $3.56B
Founded Year: 1781
Market Cap: $51.87B
Total Employees: 47,495
Headquarter: Tokyo- Japan
Stock Exchange: TYO
Takeda is a Japanese multinational biopharmaceutical company. It is one of the top 20 largest pharmaceutical companies in the world by revenue. The company is focused on metabolic disorders- gastroenterology- neurology- inflammation- as well as oncology through its independent subsidiary. With Takeda's immunology-approved drugs including Immunoglobulin and Albumin- Takeda has generated a $3.56B sale in 2020.

Immunology Segment Revenue: $4.56B
Founded Year: 1849
Market Cap: $269.75B
Total Employees: 78,500
Headquarters: New York- United States
Stock Exchange: NYSE
Pfizer is an American multinational pharmaceutical company. Pfizer develops and produces medicines and vaccines for immunology- oncology- cardiology- endocrinology- and neurology. Pfizer's immune drug- Xeljanz generated revenue of $2.43B- followed by Enbrel ($1.35B) and inflectra ($0.65B) in 2020. In Sept 2020- the US FDA approved Xeljanz (tofacitinib) for the treatment of children and adolescents aged=2yrs. with active pcJIA.

Immunology Segment Revenue: $4.60B
Founded Year: 1973
Market Cap: $128.43B
Total Employees: 100-000
Headquarters: Paris- France
Stock Exchange: EPA
Sanofi is a French multinational pharmaceutical company. The company covers seven major therapeutic areas; cardiovascular- central nervous system- diabetes- internal medicine- oncology- thrombosis- and vaccines. Dupixent and Kevzara together contributed to the $4.60B revenue of the company in the immunology segment. In November 2020- Dupixent (dupilumab) was approved by European Commission as the first and only biologic medicine for children aged 6 to 11 years with severe atopic dermatitis.

Immunology Segment Revenue: $4.86
Founded Year: 1996
Market Cap: $227.36B
Total Employees: 110,000
Headquarter: Basel- Switzerland
Stock Exchange: SWX
Novartis International AG is a Swiss multinational pharmaceutical company based in Basel- Switzerland. It is one of the largest pharmaceutical companies in the world. The company is focused on immunology- Hepatology- Dermatology- oncology- Neurology- and Ophthalmology. Novartis immune drug- Cosentyx generated revenue of $3.9B and Ilaris generated 0.87B in 2020. Cosentyx received approval and launched in the EU and US for nr-axSpA- its fourth major indication- and in August became the first treatment approved in Japan for this indication. In April- Cosentyx also became the first IL17A inhibitor approved in China for the treatment of AS. In June- Ilaris was granted a new indication in the US for active Still's disease including Adult-Onset Still's Disease (AOSD).

Immunology Segment Revenue: $7.19B
Founded Year: 1980
Market Cap: $129.45B
Total Employees: 24,300
Headquarters: California- United States
Stock Exchange: NASDAQ
Amgen is an American multinational biopharmaceutical company that focuses on CV disease- oncology- bone health- neuroscience- immunology- nephrology- and inflammation. In 2020- Enbrel- a tumor necrosis factor blocker used in the treatment of rheumatoid arthritis generated total sales of $4.9B whereas Otezla which is a PDE4 inhibitor generated a revenue of $2.1B.

Immunology Segment Revenue: $9.30B
Founded Year: 1896
Market Cap: $347.83B
Total Employees: 101,465
Headquarters: Basel- Switzerland
Stock Exchange: SWX
Roche is a Swiss multinational healthcare company. The immunology department of the company focuses on rheumatoid arthritis (RA)- systemic juvenile idiopathic arthritis- polyarticular juvenile idiopathic arthritis- and giant cell arteritis. Roche's immunotherapies include Actemra- Xolair- Esbriet- MabThera- Pulmozyme- CellCept- and others. Actemra produces the highest revenue among all with $3.23B.

Immunology Segment Revenue: $15.05B
Founded Year: 1886
Market Cap: $457.45B
Headquarter: New Jersey- United States
Total Employees: 134,500
Stock Exchange: NYSE
Johnson and Johnson is an American multinational company that focuses on multiple therapeutic areas including Cardiovascular & Metabolism- Immunology- Infectious Diseases & Vaccines- Neuroscience- Oncology- and Pulmonary Hypertension. In its immunology segment- J&J focuses on core diseases like rheumatoid arthritis- inflammatory bowel diseases- and psoriasis. In 2020- its leading immunology product- Stelara generated sales of $7.70B- followed by Remicade- Simponi- and Tremfya with the revenue of $3.74B- $2.24B- and $1.34B respectively.

Immunology Segment Revenue: $22.15B
Founded Year: 2013
Market Cap: $200.96B
Total Employees: 48,000
Headquarters: Illinois- United States
Stock Exchange: NYSE
Abbvie is an American Biopharmaceutical company focused on the development of products in immunology- oncology- virology and neuroscience- dermatology. In Dec 2020- Abbvie announced that CHMP recommended the approval of Rinvoq for the expanded use in two additional rheumatic indications: the treatment of adult patients with active psoriatic arthritis and adult.
Related Post: Top 20 Immunology Companies Based 2019 Immunology Segment Revenue
Tags

This content piece was prepared by our former Senior Editor. She had expertise in life science research and was an avid reader. For any query reach out to us at connect@pharmashots.com