Top Performing Drug of 2021 – Imbruvica (January Edition)
Active Ingredient: Ibrutinib
Dosage Forms & Strengths:
Capsules: 70 mg and 140 mg
Tablets: 140 mg, 280 mg, 420 mg, and 560 mg
Mechanism of Action: Bruton’s tyrosine kinase Inhibitor (BTKi)
First Approval: US (13 Nov 2013), EU (Oct 2014)
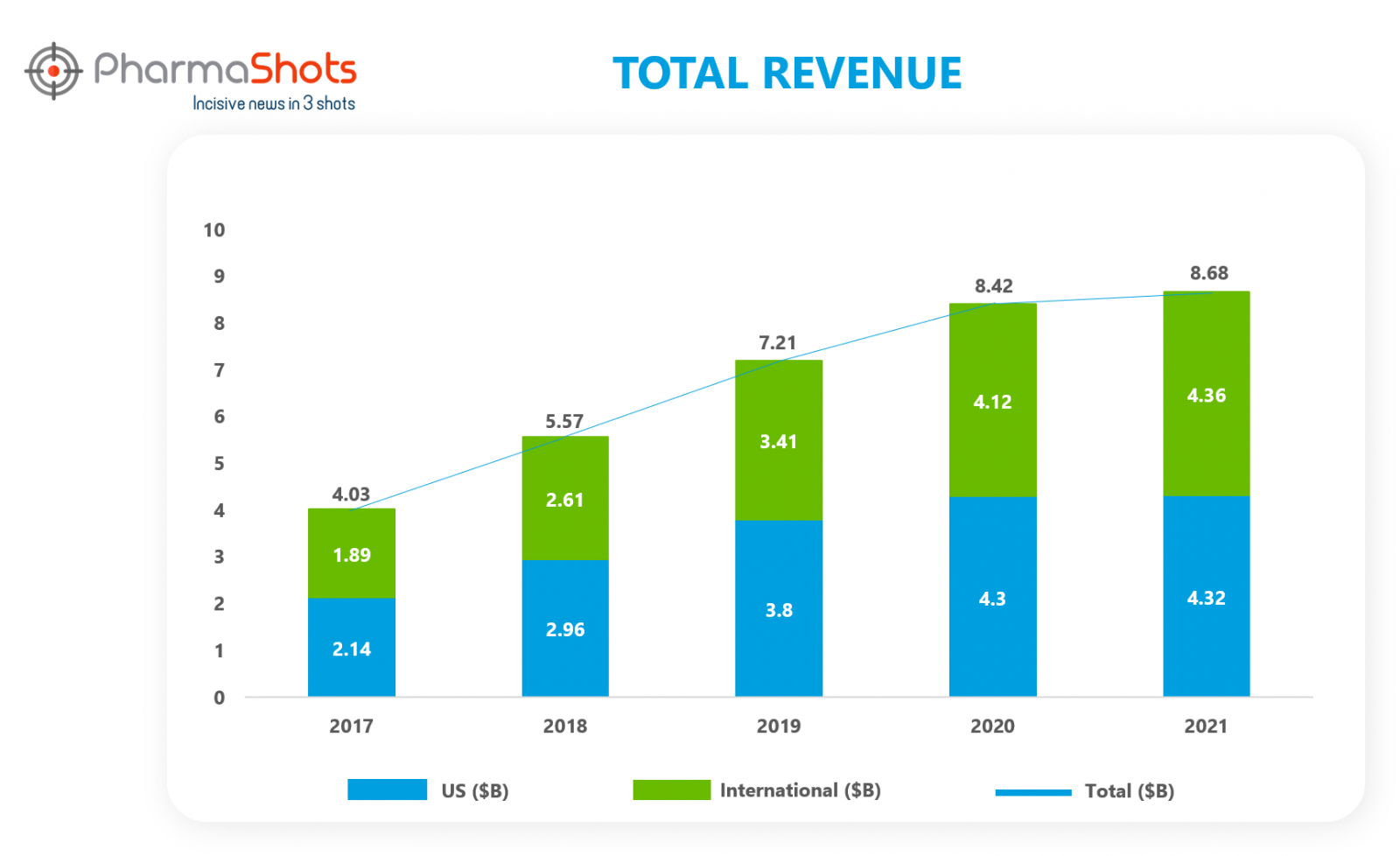
Revenue Analysis
Imbruvica is an oral therapy that inhibits a protein called Bruton’s tyrosine kinase. Imbruvica was one of the first medicines to receive US FDA approval after being granted a Breakthrough Therapy Designation and is one of the few therapies to receive four separate designations.
Imbruvica is developed jointly by AbbVie and J&J under a 2011 collaboration. According to it, Janssen has an exclusive license to commercialize Imbruvica outside of the United States and co-exclusively with AbbVie in the United States. Both parties are responsible for the development, manufacturing, and marketing of any products generated as a result of the collaboration.
PharmaShots has done a revenue analysis showing the increasing sales of Imbruvica in the past five years.
Approved Indications of Imbruvica
Imbruvica is approved and marketed in the US and Europe for various indications for the treatment of adult patients with:
- Mantle cell lymphoma (MCL) who have received at least one prior therapy.
- Accelerated approval was granted for this indication based on the overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial
- Chronic lymphocytic leukemia (CLL)/ Small lymphocytic lymphoma (SLL)
- Chronic lymphocytic leukemia (CLL)/ Small lymphocytic lymphoma (SLL) with 17p deletion
- Waldenström’s macroglobulinemia (WM)
- Marginal zone lymphoma (MZL) who require systemic therapy and have received at least one prior anti-CD20-based therapy
- Accelerated approval was granted for this indication based on overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial
- Chronic Graft versus Host Disease (cGVHD) after failure of one or more lines of systemic therapy
Clinical Trials Analysis
Imbruvica has a total of 401 trials, incl. 248 industry trials of which 231 are interventional, 15 observational & 2 are expanded access trials. The analysis of industry trials through a representation is shown below (Trials are taken as of 19 Jan 2023)

*Active trials include Recruiting; Active, Not Recruiting; Enrolling by Invitation, and suspended
*Inactive trials include Terminated; Withdrawn; Unknown Status
*Planned trials include Not, yet recruiting

Patient Assistance & Co-pay Programs for Imbruvica
Johnson & Johnson Patient Assistance Foundation
It is an independent, nonprofit organization. JJPAF gives eligible patients free prescription medicines donated by Johnson & Johnson
These patient assistance program offers free medication to people who otherwise cannot afford their medications. Patients must meet financial and other program specific criteria to be eligible for assistance. The programs cover:
- Over 35 prescription products are available to individuals who meet certain requirements and live in the United States or a U.S. Territory.
- It is free to apply and patients need to complete only one application.
- Once you meet program requirements and are approved the pateints receive their medications for up to one year. They may be able to reapply to the program annually
My AbbVie Assist
My AbbVie Assist helps patients get the medicines they need, for uninsured patients who cannot afford prescription medicines and are being treated by a licensed provider on an outpatient basis. In 2020, nearly 155,000 people were assisted through this program.
This program supports patients who:
- Are being treated by a licensed U.S. healthcare provider on an outpatient basis and prescribed an AbbVie medicine that is included in the assistance program
- Have limited or no health insurance coverage
- Demonstrate qualifying financial need
- United States residents
- Patients with commercial insurance plans requiring them to apply to myAbbVie Assist as a condition of, requirement for, or prerequisite to coverage of relevant AbbVie products commonly known as alternate funding programs, are not eligible for myAbbVie Assist
IMBRUVICA By Your Side Patient Support Program
This patient support program is designed specifically to support Imbruvica patients throughout their treatment journey. This helps patients in receiving helpful information and resources about treatment with Imbruvica. For this program, patients can sign up through online support or by calling 1-888-YourSide (1-888-968-7743)
After the patients sign up, they will receive a call from one of the Imbruvica By Your Side ambassadors. From that first call, they will:
- Provide one-on-one support throughout the treatment journey
- Connect patients to an Insurance Specialist who can provide helpful information about cost and coverage
- Provide patients with specially selected education and support materials to meet their unique needs
Further the ambassador can connect patients to an Insurance Specialist who can:
- Help them understand the insurance coverage and navigate any changes if they have federally funded insurance plans like Medicare, Medicaid, or TRICARE.
- Help eligible patients pay as little as $0 per prescription or lower your out-of-pocket costs once they are enrolled in By Your Side.
- Provide information on ways from which patients may be able to lower the cost of their prescription as well as other potential financial support options
Imbruvica Drug Interactions:
A drug-drug interaction occurs when one drug affects the activity of another. Imbruvica is known to have 422 drugs interactions along with 9 disease interactions, and 2 alcohol/food interactions. Some of them are mentioned below:
- Effect of CYP3A Inhibitors on Ibrutinib
The coadministration of Imbruvica with a strong or moderate CYP3A inhibitor may increase ibrutinib plasma concentrations. The increased amount of ibrutinib concentrations may increase the risk of drug-related toxicity.
- Effect of CYP3A Inducers on Ibrutinib
The coadministration of IMBRUVICA with strong CYP3A inducers may decrease ibrutinib concentrations.
- 5-Aminosalicylic Acid Derivatives
Imbruvica interaction with these may enhance the myelosuppressive effect of Myelosuppressive Agents
- BCG Products:
Interaction with immunosuppressants may enhance the adverse/ toxic effect of BCG Products. Also, the risk of vaccine-associated infection may be increased. Immunosuppressants may diminish the therapeutic effect of BCG Products
- COVID-19 Vaccine (Inactivated Virus):
Immunosuppressants may diminish the therapeutic effect of the COVID-19 Vaccine
- Grapefruit Juice:
Grapefruit and Seville oranges may significantly increase the blood levels of ibrutinib
- Omega-3 Fatty Acids:
It may enhance the antiplatelet effect of Ibrutinib
Imbruvica Pipeline Analysis
Imbruvica alone and in combination is being studied in multiple phases for different forms of cancers. Some of them are mentioned below:

Other Information
Patents
Patent protection is important material importance to the company for the discovery, development, manufacturing, and marketing of its products. The United States composition of matter patent covering ibrutinib is expected to expire in 2027, and the company expects no generic entry for any ibrutinib product prior to March 30, 2032, assuming pediatric exclusivity is granted.
The patent wall of Imbruvica has 353 international patents and 55 US patents. Till now, 88 patents have been granted on Imbruvica.
Patent Infringement
In March 2019, Pharmacyclics LLC and Janssen Biotech filed a patent infringement lawsuit in the United States District Court for the District of Delaware against Alvogen Pine Brook LLC and Natco Pharma Ltd. which filed an ANDA seeking approval to market generic versions of Imbruvica tablets. In August 2021, the court issued a decision holding all asserted patents infringed and valid. The judgment precludes Defendants from obtaining regulatory approval and launching until the last patent expires in 2036.
1. Johnson & Johnson 10-K reports, AbbVie 10-K reports
2. Imbruvica Prescribing Information
4. Johnson & Johnson Patient Assistance Foundation
6. IMBRUVICA By Your Side Patient Support Program
7. Imbruvica Drug Interactions, drugs.com
8. AbbVie Pipeline, J&J Pipeline
9. Patents
Related Post: Top Performing Drug of 2021 – Opdivo (December Edition)


