The US FDA New Drug Approvals in October 2024
Shots:
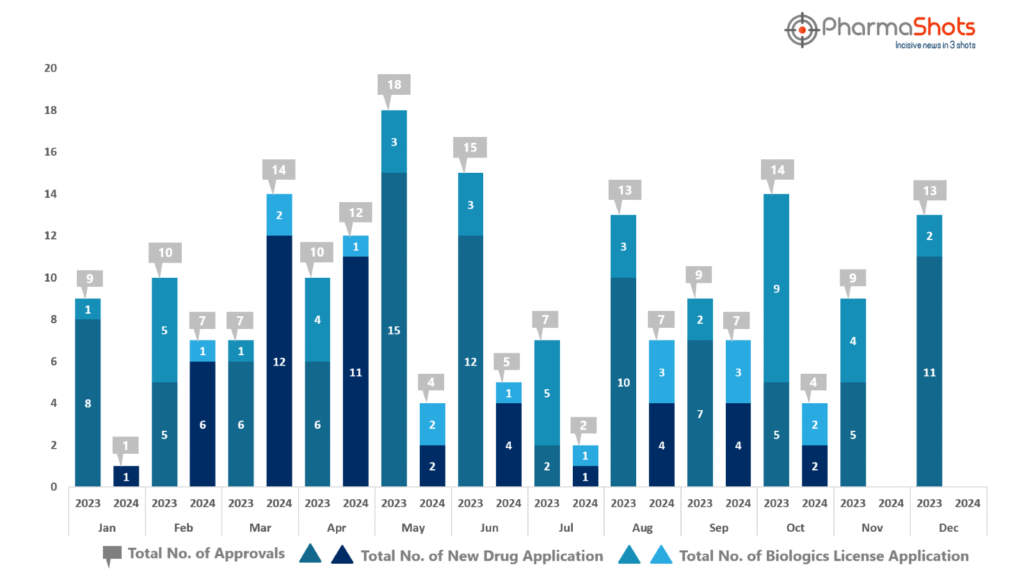
- PharmaShots has compiled a list of US FDA-approved drugs in the month of October 2024
- The US FDA has approved a total of 4 new drugs including 2 new molecular entities and 2 biologics leading to the treatment of patients and advances in the healthcare industry
- The major highlighted drugs were Pfizer’s Hympavzi for the treatment of Hemophilia A & B
Product Name: Itovebi + Ibrance & fulvestrant
Active ingredient: Inavolisib
Company: Roche
Date: Oct 10, 2024
Disease: Breast Cancer
Shots:
- The US FDA approved Itovebi + Ibrance & fulvestrant as a 1L treatment in adults with endocrine-resistant, PIK3CA-mutated, HR+ & HER2- locally advanced or metastatic breast cancer, based on the P-III (INAVO120) trial
- P-III (INAVO120) trial assessed the safety and efficacy of Itovebi + Ibrance & fulvestrant vs PBO + Ibrance & fulvestrant (n= 325) and showed a 57% reduction in disease worsening or death vs palbociclib & fulvestrant alone as 1L treatment; the trial met its 1EP (PFS) & 2EPs (ORR & CBR) with OS follow-up continuing for the next analysis
- Results from INAVO120 are also being used for submissions to other global health authorities. Additionally, the company is evaluating Itovebi in two more P-III trials (INAVO121 & INAVO122) for the same indication
Product Name: Hympavzi
Active ingredient: Marstacimab-hncq
Company: Pfizer
Date: Oct 11, 2024
Disease: Hemophilia A & B
Shots:
- The US FDA has approved Hympavzi (QW, SC) as a prophylactic treatment to prevent bleeding episodes in patients (≥12yrs.) with hemophilia A & B without FVIII & FIX inhibitors, respectively. It has also received the CHMP’s positive opinion for the same
- Results from pivotal P-III (BASIS) study, assessing Hympavzi in patients (12-75yrs.) with severe hemophilia A or mod. severe to severe hemophilia B with/without inhibitors, formed the basis of approval
- Study depicted a reduction in the annualized bleeding rate (ABR) by 35% & 92% post 12mos. with Hympavzi vs routine prophylaxis & on-demand treatment. Safety was similar to the outcomes of P-I/II study, with most common AEs being injection site reactions, headache & pruritus
3. Astellas Reports the US FDA’s Approval of Vyloy (Zolbetuximab-clzb) to Treat Advanced G/GEJ Cancer
Product Name: Vyloy
Active ingredient: Zolbetuximab-clzb
Company: Astellas
Date: Oct 18, 2024
Disease: G/GEJ Cancer
Shots:
- The US FDA has granted approval to Vyloy plus fluoropyrimidine & Pt-based CT as a 1L treatment of locally advanced unresectable or metastatic CLDN 18.2+, HER2-ve G/GEJ adenocarcinoma
- Approval was based on the P-III (SPOTLIGHT & GLOW) studies assessing Vyloy + mFOLFOX6 and Vyloy + CAPOX, respectively, vs PBO as a 1L treatment of G/GEJ cancer. Astellas partnered with Roche to use Ventana CLDN18 (43-14A) RxDx Assay for identifying eligible patients
- Results showed PFS of 24.9% (mPFS: 10.61mos. vs 8.67mos.) & OS of 25% (mOS: 18.23mos. vs 15.54mos.) in SPOTLIGHT as well as PFS of 31.3% (mPFS: 8.21mos. vs 6.8mos.) & OS of 22.9% (mOS: 14.39mos. vs 12.16mos.) in GLOW
4. Iterum Therapeutics’ Orlynvah (Oral Sulopenem) Receives the US FDA’s Approval to Treat uUTIs
Product Name: Orlynvah
Active ingredient: Sulopenem Etzadroxil & probenecid
Company: Iterum Therapeutics
Date: Oct 25, 2024
Disease: Uncomplicated Urinary Tract Infections (uUTIs)
Shots:
- The US FDA has approved Orlynvah (sulopenem etzadroxil & probenecid) to treat uUTIs in adult women, specifically addressing infections caused by E.coli, Klebsiella pneumoniae, or Proteus mirabilis, particularly when there are few or no other oral therapy available
- Approval was based on two pivotal P-III studies (SURE 1 & REASSURE) that assessed the safety & efficacy of Orlynvah vs ciprofloxacin (SURE 1) & Augmentin (REASSURE)
- The SURE 1 trial found that Orlynvah was superior to ciprofloxacin for fluoroquinolone-resistant infections, while REASSURE demonstrated both non-inferiority & statistical superiority to Augmentin among patients susceptible to that antibiotic. Both trials indicated well-tolerability
Related Post: Insights+: The US FDA New Drug Approvals in September 2024





