AstraZeneca at ACR 2023: Andrew Menzies-Gow Highlights Data from the MANDARA Study
Shots:
- Andrew Menzies-Gow, in a stimulating conversation with PharmaShots, shares the insights from the Phase III MANDARA study that evaluates Fasenra (benralizumab) vs. mepolizumab for patients with relapsing or refractory EGPA
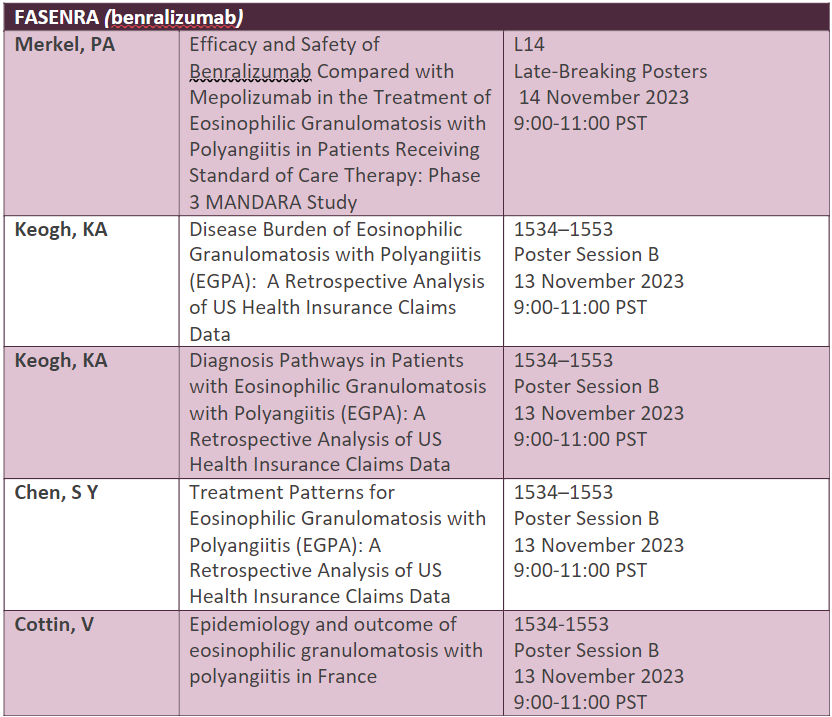
- Five out of the 18 abstracts presented by AstraZeneca at ACR were from the MANDARA study. Apart from the positive endpoints of the MANDARA study data on secondary endpoints were also shared at ACR
- AstraZeneca is exploring benralizumab’s potential in other diseases that may be driven by eosinophilic inflammation
Saurabh: As it’s observed that Fasenra (benralizumab) is already under development so what are the updates regarding the same?
Andrew: We’re exploring benralizumab’s potential in other diseases that may be driven by eosinophilic inflammation.
Eosinophilic inflammation may be an important driver in hyper-eosinophilic syndrome (HES), chronic rhinosinusitis with nasal polyps (CRSwNP), and some patients with chronic obstructive pulmonary disease (COPD). [For more on benralizumab’s clinical development program].
MANDARA is the first Phase III head-to-head trial of biologics in eosinophilic granulomatosis with polyangiitis (EGPA), comparing the efficacy of Fasenra to mepolizumab, and the first trial to demonstrate that a biologic medicine given in a single injection every four weeks could help patients achieve remission at rates comparable to mepolizumab’s three injections every 4 weeks. This suggests that benralizumab may be a valuable future treatment option for EGPA.
Our ambition is to build on the position benralizumab has already achieved in severe eosinophilic asthma (SEA) to be the first-line, first-choice biologic for eosinophil-driven diseases.
In addition, we’re sharing the positive late-breaking full results from the Fasenra MANDARA Phase III trial, the first head-to-head trial of biologics to treat eosinophilic granulomatosis with polyangiitis (EGPA).
Full abstract list below:
Saurabh: P-III study (MANDARA) is evaluating benralizumab in comparison with mepolizumab, what is its design study & is it more effective & safer than mepolizumab?
Andrew: MANDARA was a randomized, double-blind, double-dummy, active-controlled, parallel-group, multicenter 52-week Phase III trial that compared the efficacy and safety of Fasenra to mepolizumab in adult patients with relapsing or refractory EGPA.
In the blinded trial, 140 patients were randomized 1:1 (70 per treatment group) to receive either a single 30mg subcutaneous injection of Fasenra or three separate 100mg subcutaneous injections of mepolizumab once every four weeks.
MANDARA was designed as a non-inferiority trial and was not powered to show superiority between Fasenra and mepolizumab.
MANDARA achieved its primary endpoint, demonstrating that benralizumab was non-inferior compared to mepolizumab on remission rate. Remission was defined as Birmingham Vasculitis Activity Score (BVAS)=0 and OGC dose less than or equal to 4mg/day. The data showed that 59.2% of benralizumab-treated patients achieved remission, compared to 56.5% for those treated with mepolizumab (p=0.7278).
- Data on secondary endpoints were also shared at ACR, including:
- Oral glucocorticoids (OGCs) tapering: 41.4% of patients on benralizumab vs. 25.8% on mepolizumab were fully tapered off from OGCs, a nominally significant difference (95% CI: 0.67, 30.71; p=0.0406).
- 42.1% of benralizumab-treated patients achieved remission by week 24 and maintained remission until week 52 vs 36.5% on mepolizumab (95% CI: -9.30, 20.37; p=0.4643)
- At 30%, the proportion of patients who relapsed was the same for benralizumab and mepolizumab.
- 62.5% of benralizumab-treated patients achieved complete response vs. 54.6% of mepolizumab patients (95% CI: -7.32, 23.12; p=0.3088) defined as achieving remission at any time during the double-blind period, ≥50% reduction in daily OGC dose during weeks 49-52 and being relapse-free during the double-blind period.
Image Source: Canva
About the Author:

Andrew Menzies-Gow Bop
Andrew Menzies-Gow is the Global Medical Head for Respiratory Biologics at AstraZeneca. Prior to joining the company in January 2023, Andrew was the clinical and research lead for the adult severe asthma service and Director of the Lung Division at the Royal Brompton & Harefield Hospitals, London, UK. Andrew was a Professor of Practice (Respiratory Medicine) at Imperial College, London, UK, where his research interests focused on novel therapies for severe asthma and was the National Clinical Director for Respiratory Disease at NHS England.
Related Post: AstraZeneca at ACR 2023: Micki Hultquist Shares Post-hoc Analysis of Saphnelo

