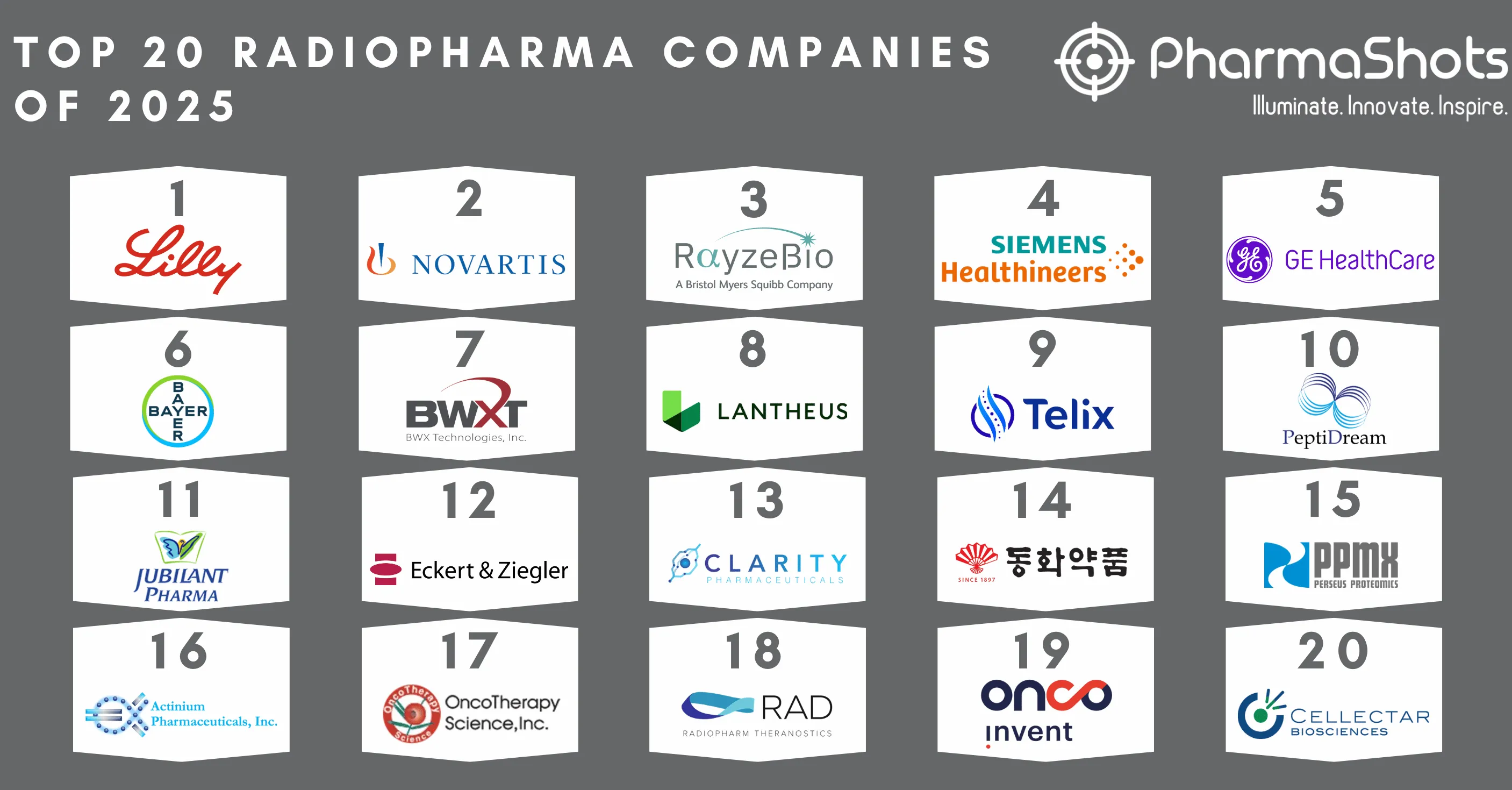
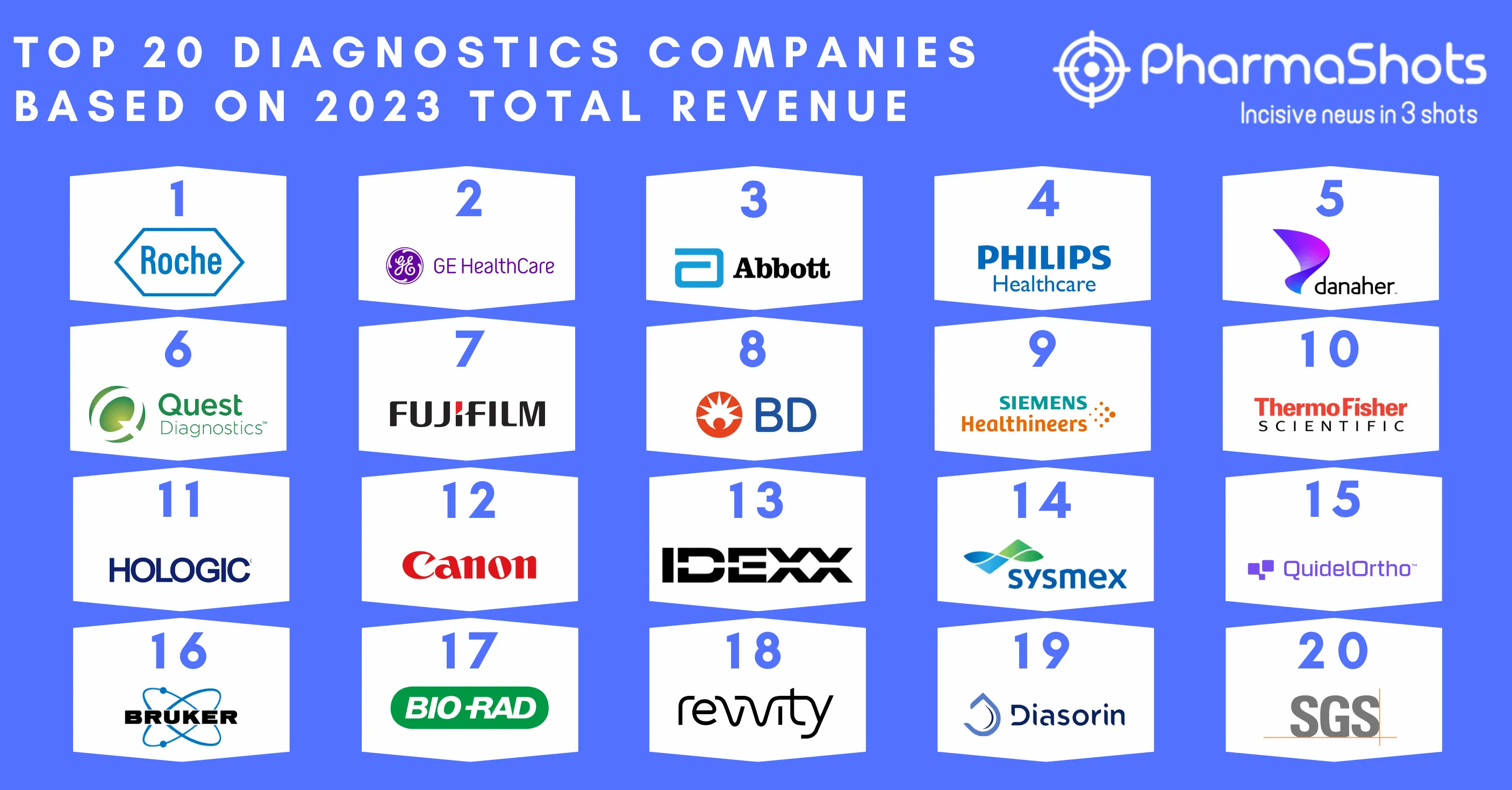
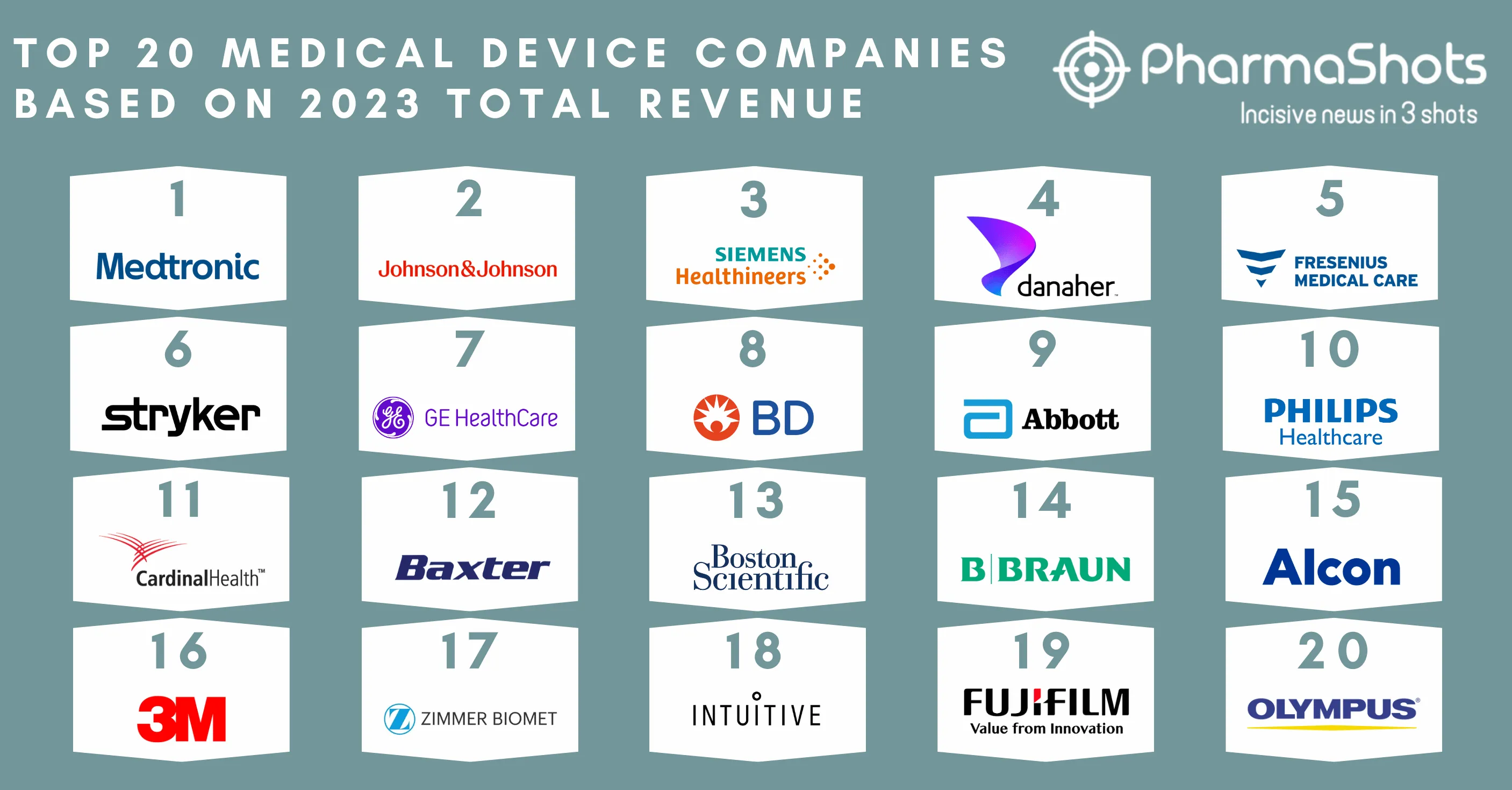
Siemens Healthineers Receives the US FDA Approval for Biograph Vision.X PET/CT Scanner
Shots:
- The approval of Biograph Vision.X is a positron emission tomography/computed tomography with a time of flight (TOF) of 178ps
- The Biograph Vision.X scanner utilizes Optiso UDR technology with SiPMs and tiny 3.2 mm by 3.2 mm LSO crystals for enhanced spatial resolution. It achieves an industry-best temporal resolution of 178 ps & a 48 mm³ volumetric resolution. The fully covered LSO crystals by SiPMs result in a 20% performance gain (TOF gain), reducing radiotracer costs, increasing patient throughput, & lowering radiation exposure
- The next-generation Biograph Vision.X PET/CT Scanner builds upon the proven performance of the Biograph Vision scanner family and the AIDAN intelligent imaging platform
Ref: Siemens | Image: Siemens
Related News:- Siemens Healthineers’s CLINITEST COVID-19 Antigen Test Receives the US FDA's EUA for the Prevention of COVID-19
PharmaShots! Your go-to media platform for customized news ranging for multiple indications. For more information connect with us at connect@pharmashots.com
Click here to read the full press release

Kritika is a content writer at PharmaShots. She is interested in covering recent innovations from the pharma & MedTech industry. She covers news related to Product approvals, clinical trial results, and updates. She can be contacted at connect@pharmashots.com.