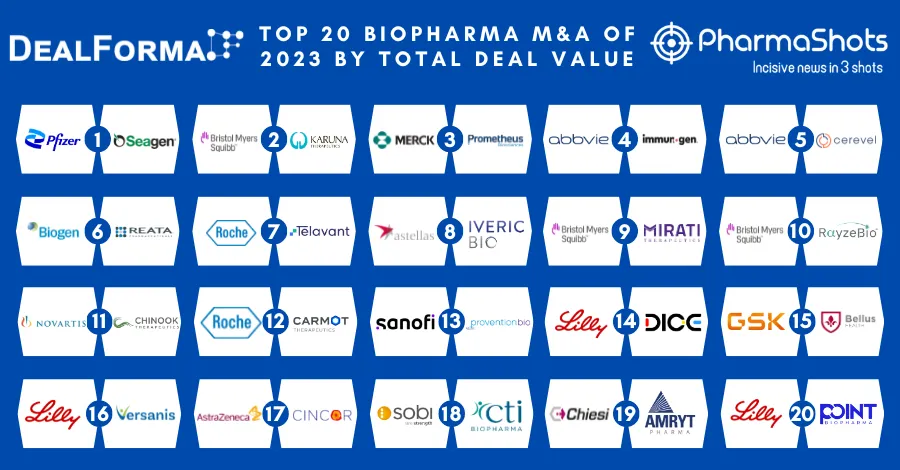
Chiesi’s Elfabrio (pegunigalsidase alfa) Receives NICE Recommendation for Fabry Disease
Shots:
- The NICE has recommended Chiesi’s Elfabrio as a long-term option for Fabry disease among adult patients with a confirmed diagnosis in England and Wales. Elfabrio, a novel enzyme replacement therapy is administered via IV infusion, q2w
- The recommendation was based on the P-III trials which showed that Elfabrio was well tolerated and was similarly clinically effective and as tolerable as the current treatments, agalsidase alfa and migalastat
- The therapy was found to be a more cost-effective option over other ERTs & migalastat. Pegunigalsidase alfa was approved in the EU, Northern Ireland & Great Britain for long-term ERT in adult patients who have been confirmed to have the disorder & in the US for confirmed Fabry disease in adults
Ref: PharmaTime | Image: Cheisi
PharmaShots! Your go-to media platform for customized news ranging for multiple indications. For more information connect with us at connect@pharmashots.com
Click here to read the full press release

Neha is a Senior Editor at PharmaShots. She is passionate and very enthusiastic about recent updates and developments in the life sciences and pharma industry. She covers Biopharma, MedTech, and Digital health segments along with different reports at PharmaShots. She can be contacted at connect@pharmashots.com.