
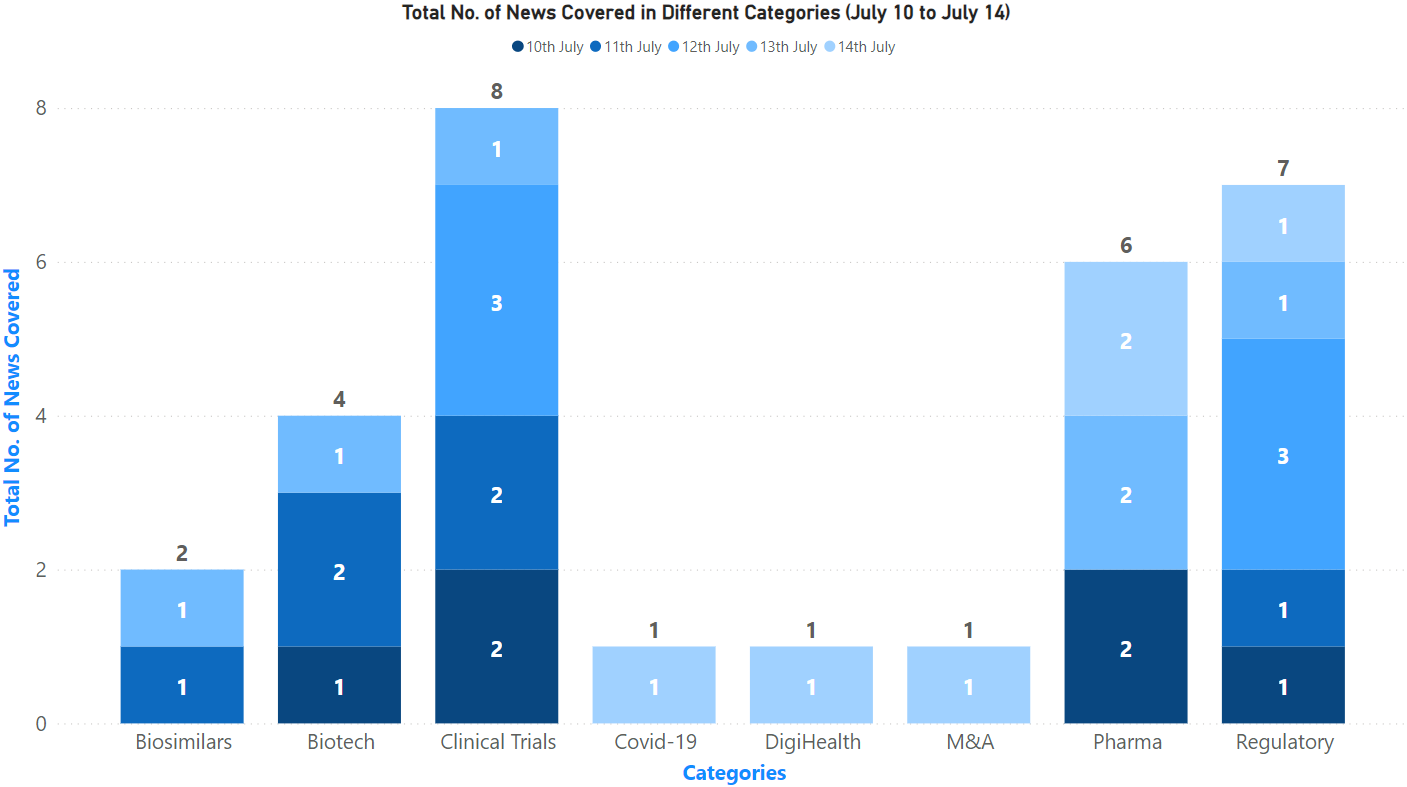
PharmaShots Weekly Snapshots (July 10 – 14, 2023)
This week PharmaShots’ news was all about the updates on clinical trials, regulatory developments, biosimilars, biotech, and pharma. Check out our full report below:

- Roche collaborated with KSQ on their novel inhibitor of USP1 as a new treatment option for patients with cancers
Read more: KSQ
- Astellas signed a ~$962M deal with 4D Molecular Therapeutics for rare ophthalmic targets using 4DMT's Intravitreal R100 vector
Read more: Astellas
- Fulcrum and CAMP4 collaborated to develop rare blood disorder therapies
Read more: Fulcrum
- BeiGene and DualityBio partnered on differentiated antibody-drug conjugate therapy for Solid Tumors while DualityBio will lead the preclinical research activities and support IND filings
Read more: BeiGene

- Acadia & Neuren expanded the license agreement to acquire Ex-North American rights to Trofinetide and global rights to NNZ-2591
Read more: Acadia
- InnoCare and ArriVent collaborated to evaluate ICP-189 + furmonertinib for NSCLC and will provide the benefit to global patients early
Read more: InnoCare
- LianBio collaborated with AstraZeneca to evaluate NSCLC combination therapy in the P-I trial which is expected to initiate in H2’23
Read more: LionBio
- Gilead partnered with CHAI and the Pentato to advance the development of dispersible pediatric therapies and improved HIV treatment in children
Read more: Gilead
- NANOBIOTIX signed a license agreement with Janssen to co-develop and commercialize first-in-class radioenhancer NBTXR3
Read more: NANOBIOTIX
- RemeGen and Innovent collaborated to conduct clinical trials for RC88 and RC108 with Tyvyt (sintilimab) in patients with advanced solid tumors
Read more: RemeGen

- The US FDA, EMA, and MHRA accepted Dr. Reddy's rituximab biosimilar candidate DRL_RI for substantive review
Read more: Dr. Reddy's
- Aurobindo Pharma subsidiary CuraTeQ in pact to commercialize biosimilar of BioFactura’s recombinant monoclonal antibody Stelara
Read more: Aurobindo Pharma

- Roche highlighted P-III trial (OCARINA II) results for investigational SC formulation of ocrelizumab which was found non-inferior to IV infusion in MS
Read more: Roche
- Daré Bioscience reported additional results from an exploratory P-IIb Study (RESPOND) for Sildenafil with improvement in arousal and orgasm, as well as sexual desire for female sexual arousal disorder
Read more: Dare Bioscience
- ADC paused Zynlonta trial enrolment after observing seven deaths and five adverse events for patients with previously untreated DLBCL
Read more: ADC
- Incyte's Opzelura meets the positive results in the P-III study and induced treatment success in a higher proportion of children with AD
Read more: Incyte
- Ionis reported 85-week results from the P-III (NEURO-TTRansform) study of Eplontersen with improvement in in measures of neuropathy disease
Read more: Ionis
- HUTCHMED dosed the first patient in the P-I study of HMPL-415 for advanced malignant solid tumors in China
Read more: HUTCHMED
- Axsome Therapeutics dosed the first patient in an ADHD therapy trial
Read more: Axsome Therapeutics
- BMS highlighted the sub-study results of the P-III trial (CheckMate -901) for Opdivo in urothelial carcinoma and met the dual 1EPs of OS & PFS at the final analysis
Read more: BMS

- The NMPA has accepted the NDA of Ardelyx’s Tenapanor to treat Hyperphosphatemia in China
Read more: Ardelyx
- The NMPA approved Daiichi Sankyo and Astrazeneca’s Enhertu for HER2 low metastatic breast cancer, based on the P-III trial (DESTINY-Breast04) results
Read more: Daiichi Sankyo
- The US FDA accepted BeiGene’s sNDA of Brukinsa + obinutuzumab for follicular lymphoma therapy, based on the P-II study (ROSEWOOD) which showed an efficacy benefit
Read more: BeiGene
- TFDA granted accelerated marketing approval for Zepzelca in adult patients with metastatic SCLC with disease progression on or after Pt-based CT
Read more: PharmaMar
- The NMPA accepted the Junshi Biosciences’ sNDA of toripalimab + axitinib as 1L treatment of unresectable or metastatic renal cell carcinoma
Read more: Junshi Bioscience
- Roche gets EC nod for Columvi as a fixed course induced early and long-lasting complete responses for patients with relapsed or refractory diffuse large b-cell lymphoma
Read more: Roche
- Ichnos Sciences’ ISB 2001 receives the US FDA’s orphan drug designation for the treatment of multiple myeloma
Read more: Ichnos sciences

- Korro Bio entered into a definitive merger agreement with Frequency Therapeutics to advance RNA editing programs
Read more: Korro Bio

- Aerin Medical highlighted two-year results supported Aerin Medical treatment for nasal airway obstruction
Read more: Aerin Medical

- Gilead received the US FDA’s approval of Veklury (remdesivir) sNDA for COVID-19 in patients with Severe Renal Impairment, based on the results from a P-I study (GS-US-540-9015) & the P-III trial (REDPINE)
Related Post: PharmaShots Weekly Snapshots (July 03 – 07, 2023)














