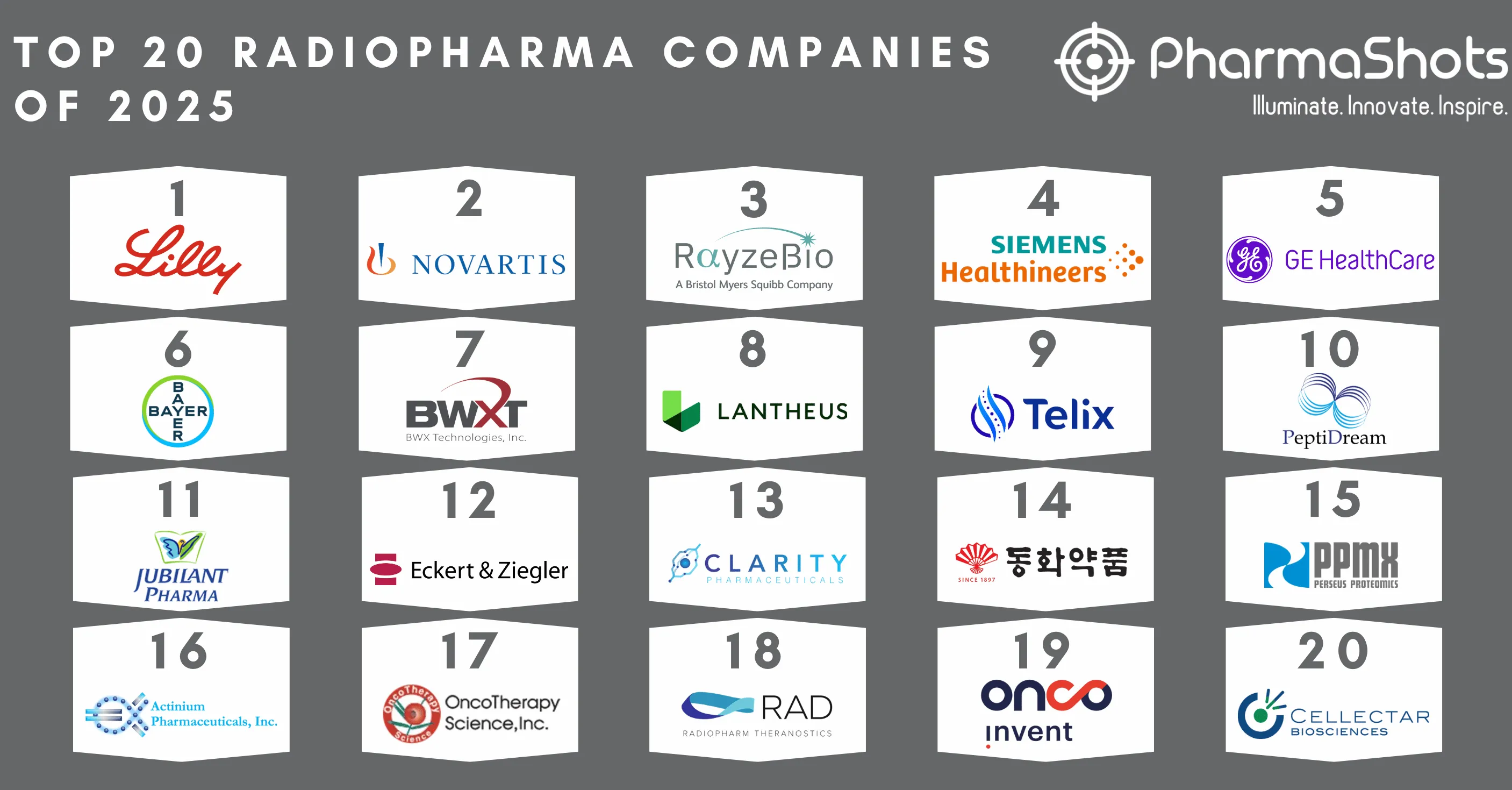
Top 20 Biopharma Deal Terminations of 2022 Based on Total Deal Value
Shots:
- Post M&A deals, companies often cannot sail through a few key aspects of the partnership, such as limited ownership, exorbitant recovery costs, and poor integration process, leading to deal terminations. The improperly executed expansion program also contributes significantly to the failed partnership
- To tackle such scenarios, companies are cautious enough to make backup plans beforehand to handle such instances without incurring a major loss. In 2022, the deal between Biogen and Catalyst Biosciences was the largest deal termination followed by Amicus’ deal with ARYA Sciences and Amgen’s deal with Molecular Partners
- With the invaluable insights of DealForma, PharmaShots has prepared a list of the Top 20 Biopharma Deal Terminations of the year 2022 based on the total deal value

Biogen Terminated its License and Collaboration Agreement with Sangamo
- In Jan 2014, Sangamo entered into an exclusive global collaboration and license agreement with Biogen. Sangamo granted Biogen the worldwide rights to develop and commercialize therapeutics for hemoglobinopathies including sickle cell disease and beta-thalassemia using Sangamo’s zinc-finger ZFN genome-editing technology. As per the agreement, Sangamo was responsible for all R&D activities through the first clinical proof of concept trial in beta-thalassemia whereas both companies were responsible for an IND submission for sickle cell disease
- On the other hand, Biogen was given the responsibility for subsequent worldwide clinical development and commercialization of products developed under the agreement while Sangamo had the option to co-promote any licensed product to treat sickle cell disease and beta-thalassemia in the US. Under the transaction, Sangamo received an up front payment of $20M and was eligible to receive development, regulatory, and sales milestone payments worth $293.8M including a P-I milestone payment of $7.5M for each of the beta-thalassemia and sickle cell disease programs along with double-digit royalties. SAR445136 was the collaborative drug produced under this agreement. Later in the year 2016, Biogen spun off its hemophilia business into a company called Bioverativ
- In Nov 2016, Sangamo reported the transfer of its Biogen agreement to Bioverativ. In Jan 2018, Bioverativ got acquired by Sanofi which then in Mar 2021 out-licensed its sickle cell programs (obtained from Bioverativ) to Global Blood Therapeutics. Under the Sanofi and Global Blood Therapeutics agreement, Sanofi received an undisclosed up front payment and was eligible to receive up to $353M in milestone payments, plus single-digit tiered royalties. Later in Jan 2022, Sanofi reported the termination of its deal with Sangamo and thereby transferred all rights to P-I/II SAR445136 back to Sangamo

Biogen Terminates its Global License and Collaboration Agreement with Catalyst Biosciences
- In Dec 2019, Catalyst Biosciences collaborated with Biogen under which Biogen received the exclusive, worldwide rights to develop and commercialize Catalyst’s CB-2782 and other anti-C3 proteases for the treatment of geographic atrophy (GA) associated dry age-related macular degeneration (dry AMD)
- Through this transaction, Catalyst received $15M up front (in Jan 2020) and would have been eligible for undisclosed R&D funding, up to $340M in milestones along with low double-digit tiered royalties
- In Mar 2022, Biogen returned the rights for the future development of CB-2782 and thereby ending the collaborative agreement between the two companies

Amicus Therapeutics Terminated its Business Combination Agreement with ARYA Sciences Acquisition Corp. IV
- In Sep 2021, Amicus Therapeutics entered into a definitive business combination agreement with AYRA Sciences under the terms of which, Amicus’ gene therapy was to be acquired by ARYA to form a combined company called Caritas Therapeutics
- Amicus was the largest shareholder in the combined company with a ~36% ownership stake. As per the terms of the agreement, the combined company received cash proceeds of $150M held in ARYA IV’s trust account and $200M as PIPE financing
- Additionally, Amicus invested $50M for additional stakes in the combined company and the combined company was expected to receive proceeds of ~$400M at the closing of the transactions. Later in Feb 2022, Caritas (the combined company) terminated its SPAC agreement with ARYA due to unfavorable market conditions

Amgen Terminates its Clinical Development and Commercialization Agreement with Molecular Partners
- In Dec 2018, both Amgen and Molecular Partners entered into a license agreement to clinically develop and commercialize MP0310 for the treatment of cancer. Under the terms of the transaction, Molecular Partners and Amgen were to jointly evaluate MP0310 in combination with Amgen’s oncology pipeline products including its BiTE (bispecific T cell engager) molecules
- Additionally, Molecular Partners retained certain rights to develop and commercialize its DARPin pipeline products in combination with MP0310. As per the agreement, Molecular Partners received an upfront payment of $50M and would have been eligible for up to $497M in development, regulatory, and commercial milestones plus up to high teens double-digit tiered royalties
- Molecular Partners were primarily responsible for performing clinical development, manufacturing, and regulatory activity in the first clinical phase whereas Amgen was responsible for the development costs for additional clinical trials. In Apr 2022, Molecular Partners reported Amgen’s decision to return the global rights of MP0310 following a strategic pipeline review

Sanofi Terminated its Clinical Development and Commercialization Agreement with Revolution Medicines
- In Jul 2018, Sanofi and Revolution Medicines entered an exclusive global partnership to co-develop and commercialize SHP2 inhibitors including RMC-4630 for the treatment of patients with cancers carrying certain mutations including NSLC
- As per the deal terms, Revolution will continue to lead research and early clinical development whereas Sanofi will be responsible for the later development activities. Both companies entered a 50/50 profit and loss share agreement in the US. Upon agreement, Revolution received an up front payment of $50M and would have been eligible to receive more than $500M in development and regulatory milestones along with tiered royalties reaching mid-double digits on sales in other markets
- Additionally, Revolution could have also received an option to co-promote any approved products targeting SHP2 in the US. In Dec 2022, the agreement between Sanofi and Revolution Medicines was terminated.

Cytovia Therapeutics Terminated its Business Combination Agreement with Isleworth Healthcare
- In Apr 2022, Cytovia Holdings entered into a definitive business combination agreement with Isleworth to form a combined company named Cytovia Therapeutics. The combined company would continue Cytovia’s operation along with developing and manufacturing complementary NK cell and NK engager antibody platforms
- The transaction valued the combined company at a pro forma equity value of $602M at a $10 per share price with no redemptions. Cytovia shareholders received $227M in gross proceeds including $207M in cash from Isleworth's trust account and received $20M in PIPE financing by issuing 2,600,000 shares of Isleworth Common Stock
- Isleworth stockholders were to receive certain payments in two tranches upon achieving $15.00 and $20.00 per share in the first and second years. Later in Jun 2022, Cytovia terminated its merger agreement with Isleworth.

Eleusis Holdings Terminated its Business Combination Agreement with Silver Spike Acquisition Corp
- In Jan 2022, Eleusis Holdings entered into a definitive business combination agreement with Silver Spike. Under the agreement, both companies jointly formed a combined company called Eleusis Inc. As per the transactions, the combined company had an enterprise value of $446M
- The shareholders at Eleusis received $350M in the combined company at $10 per share and were eligible to receive earn-out shares of up to 14% of the pro forma value. Out of the earn-out shares, 20% of the shares were vested if 20 days weighted average in 30 days exceeded $12.5 per share, 30% were vested if the share price exceeded $15 per share, and 50% were vested if the share exceeded $17.5 per share
- Additionally, Eleusis was expected to receive $287.5M in gross proceeds from Spike’s trust account whereas Spike was expected to receive more than $50M from external finance. Eleusis would retain ~49% of the combined company with no redemptions by Spike’s shareholders. The merger between Eleusis and Sliver Spike was terminated in Jun 2022

Global Cord Blood Corporation Terminated its Acquisition of Cellenkos
- In Jul 2018, Global Cord Blood Corporation (GCBC) reported the acquisition of Cellenkos to get hold of its lead products including Phase I CK-0802 for COVID-19 and other lung diseases, Phase I CK-0801 for Bone marrow disorders, and Preclinical/IND CK-0805 for Graft vs Host disease
- As per the terms of the agreement, shareholders of Cellenkos received 65,700,000 GCBC ordinary shares at $0.0001 par value per share and 36,112,267 ordinary shares on a fully diluted basis. Additionally, GCBC owned 95% of Cellenkos’ outstanding equity interest while the remaining 5% was to be held by the GMPM shareholders
- At closing, Cellenkos was expected to receive $664M in cash and 125M new shares, on an as converted and fully diluted basis, valued at $11 per share. Later in Jun 2022, GCBC announced it will terminate the acquisition of Cellenkos

Bayer Terminates its Clinical Development and Commercialization Agreement with Atara Biotherapeutics
- In Dec 2020, Bayer partnered with Atara Biotherapeutics for the exclusive worldwide license to research, develop, manufacture, and commercialize mesothelin-directed CAR T-cell therapies for the treatment of solid tumors including NSCLC. The agreement includes the development of ATA3271 & ATA2271 by utilizing Atara's Epstein-Barr Virus (EBV) T Cell platform. According to the agreement, Atara was held responsible to lead IND-enabling studies and process development for ATA3271 while Bayer was responsible for submitting the IND and subsequent clinical development and commercialization at their own costs. In terms of ATA2271, Atara retained the responsibility to continue the ongoing P-I study following which Bayer would have been held responsible for the further development of ATA2271 at its own costs
- As per the transaction, Atara received an up front payment of $45M in cash and a $15M up front reimbursement payment for certain research and process development activities to be performed by Atara. Atara also received up to $5M for additional specified translational activities and would have been eligible to receive $610M in milestone payments along with low-double-digit tiered royalties. Later in Mar 2021, both companies expanded their collaboration agreement by entering a manufacturing, supply, pharmacovigilance, quality, and technology transfer agreement. The technology transfer agreement highlights the transfer of the ATA3271 manufacturing process for which Atara invoiced Bayer $3.1M (20%) out of $15.3M in the second quarter of 2021 and $6.1M (40%) in Jan 2022. The remaining 40% of the fee was to be transferred as 20% in Jan 2023 and 20% upon the technology transfer completion
- Additionally, under the manufacturing and supply agreement, Atara was eligible to receive $13.1M of supply price out of which $6.6M was received in the second quarter of 2021 and the remaining was to be received upon the release of the order. In May 2022, Bayer reported the intent to terminate its license agreement with Atara. Thereby in Sep 2022, prior to the termination effective date, Bayer paid Atara $4.2M in return for certain activities performed by Atara.

Neurimmune and Alexion, AstraZeneca’s Rare Disease Group, Terminates their Development and Commercialization Agreement
- In Jan 2022, Alexion received the exclusive, worldwide license to develop, manufacture, and commercialize Neurimmune’s ATTR clinical asset NI006. NI006 is a mAb developed using Neurimmune's Reverse Translational Medicine technology to target misfolded transthyretin for the treatment of transthyretin amyloid cardiomyopathy (ATTR-CM)
- As per the agreement, Neurimmune was held responsible for the completion of the ongoing P-Ib clinical trial following which Alexion would have been responsible for all further development and commercialization activities
- From this transaction, Neurimmune received an up front of $30M and would have been eligible for up to $730M in development, regulatory, and commercial milestones in addition to low-to-mid teen royalties. In Mar 2022, Neurimmune reported the closing of its collaboration with Alexion

Allergan and Exicure Mutually Terminated their R&D and Option to License Agreement
- In Nov 2019, Exicure granted Allergan an option to license the exclusive worldwide rights to develop and commercialize SNA-based therapeutics discovered through two collaborative programs by using Exicure's SNA technology for the treatment of hair loss disorders
- As per the terms of the agreement, Exicure received an up front payment of $25M and if exercised, Allergan would have paid an exercise fee equal to $10M per program. If Allergan had elected to extend the initial option exercise period, then it would have paid an additional fee of $10M and if Allergan elects to exercise its option during the extended option exercise period, it would have to pay Exicure the option fee of $15M
- Additionally, Exicure would have been eligible to receive $55M in development, $132.5M in regulatory, and $175 in commercial milestones for each program along with mid-single digits to mid-teens tiered royalties. In May 2020, Allergan got acquired by AbbVie in accordance with which Allergan granted AbbVie an exclusive option to license these SNA-based therapeutics. Later in Dec 2022, Both Allergan and Exicure mutually terminated the agreement

Everest Medicines Terminated its Single-Asset License Agreement with Immunomedics
- In Apr 2019, Immunomedics agreed to grant Everest Medicines the exclusive rights to develop and commercialize its antibody-drug conjugate sacituzumab govitecan (IMMU-132/Trodelvy) across Greater China, South Korea, and certain Southeast Asian territories for the treatment of cancer. Everest was held responsible for the development of sacituzumab govitecan in various global and local indications including solid tumors across its territory
- As per the terms of the agreement, Immunomedics received an up front payment of $65M and would have been eligible to receive $60M in regulatory milestones upon the approval of sacituzumab govitecan by the US FDA to treat mTNBC. Additionally, Immunomedics was also eligible to receive $710M in development and commercial milestones along with escalating mid-teens tiered royalties. In Oct 2020, Immunomedics got acquired by Gilead following which Everest returned the development and commercialization rights for sacituzumab govitecan to Immunomedics's holding company Gilead
- Later in 2022, the agreement between Everest and Immunomedics was restated thereby terminating the 2019 agreement between the two companies. Based on the restated agreement between Everest and Gilead, Everest received an upfront payment of $280M and is eligible to receive $175M in regulatory and commercial milestones. Following the Gilead agreement, Everest was released from the payment obligation for up to $710M in milestone payment as per its initial agreement with Immunomedics

Sio Gene Therapies Terminated its Licensing Agreement with Oxford BioMedica
- In Jun 2018, Oxford BioMedica granted Sio Gene Therapies (then Axovant Sciences) the exclusive worldwide license to develop and commercialize AXO-Lenti-PD (previously named OXB-102) gene therapy for the treatment of Parkinson’s disease by utilizing the LentiVector platform
- Under the terms of the agreement, Oxford BioMedica received an up front payment of $30M including a $5M pre-payment for manufacturing activities related to AXO-Lenti-PD. Oxford BioMedica would have been eligible to receive $55M in development milestones and $757.5M in regulatory and sales milestones
- Oxford was also eligible to receive tiered royalty from 7-10%. In Jan 2022, Sio Gene Therapies reported its intent to return AXO-Lenti-PD’s global rights to Oxford BioMedica

GSK Terminated its Strategic Collaboration and Licensing Agreement with Adaptimmune Therapeutics
- In Jun 2014, Adaptimmune entered into an agreement with GSK for the development and commercialization of its T-cell therapy targeting NY-ESO-1. As per the agreement signed, Adaptimmune was responsible to discover up to 5 products under its NY-ESO TCR program whereas GSK was given the option to take over the complete development and commercialization of those products. For the initial 7yrs., Adaptimmune could generate payments more than $350M. In Feb 2016, both Adaptimmune and GSK agreed upon expanding the deal to include GSK3377794 for the treatment of synovial sarcoma. Adaptimmune could also initiate up to 8 POC studies with combinations of checkpoint inhibitors
- GSK agreed to fund the pivotal studies through milestone-based payments and had the right to nominate up to 4 additional targets eligible for milestones with Adaptimmune. As per the 2016 expansion, Adaptimmune was eligible for up to $500M in payments along with undisclosed amounts received previously if GSK licenses and develops NY-ESO for more than one indication and more than one HLA. Adaptimmune was also eligible for additional undisclosed tiered sales milestones and undisclosed mid-single to low double-digit royalties on global sales. Another deal expansion between Adaptimmune and GSK took place in Sep 2017 for the inclusion of the NY-ESO SPEAR T-cell therapy program. GSK could also nominate two additional programs
- As per the 2017 expansion, Adaptimmune was expected to receive up to $61M in near-term payments including $23M as a development milestone and $38M in option payments. Upon commencement of the license in 2018, the previous performance obligation relating to the NY-ESO license was recognized. In 2019, GSK nominated its third target under the collaboration for which Adaptimmune received $3.2M. In Oct 2022, GSK terminated the deal by returning Adaptimmune the rights for PRAME and NY-ESO cell therapy program. GSK will be responsible to provide P-II potential registrational trial data along with all development costs

Janssen Pharmaceuticals Terminated Collaboration and Licensing Agreement with Bavarian Nordic
- In Jul 2017, under its collaboration, Bavarian granted Janssen the exclusive worldwide rights to its MVA-BN technology for two additional programs targeting vaccines against HBV and HIV-1
- As per the transaction, Bavarian received an up front payment of $10M and $33M in up front equity investment. Additionally, Bavarian was also eligible to receive a total of $836M in development, regulatory, and commercial milestones in addition to tiered royalties
- Later in May 2022, Janssen reported that its collaboration agreement with Bavarian to develop HBV and HPV vaccines had been terminated, but Janssen would still be responsible for the development of HIV and Ebola vaccines.

Ipsen Terminated its Development and Commercialization Agreement with Exicure
- In Feb 2021, Exicure signed an agreement with Ipsen under which Ipsen was granted an exclusive worldwide license to develop and commercialize spherical nucleic acids (SNA)-based therapeutics for the treatment of Huntington's Syndrome and Angelman Syndrome
- Exicure was held responsible for the discovery and certain pre-clinical development activities and Ipsen was held responsible for further development and commercialization of the licensed products
- As per the terms of the agreement, Exicure received $20M up front and was eligible to receive $1B in option exercise fees and milestone payments plus royalties if Ipsen opts into both programs. In Dec 2022, Exicure reported the termination of its license agreement with Ipsen

Pfizer Terminated its License Agreement with Ionis Pharmaceuticals
- In Oct 2019, Akcea, an affiliate of Ionis Pharmaceuticals, entered into an exclusive worldwide license agreement with Pfizer for the development of AKCEA-ANGPTL3-LRx to treat people with cardiovascular and metabolic diseases
- As per the agreement, Akcea conducted a P-II evaluation of AKCEA-ANGPTL3-LRx for the treatment of NAFLD whereas Pfizer was held responsible for all development and regulatory activities following the completion of the P-II clinical trial. Akcea retained the rights to co-commercialize AKCEA-ANGPTL3-LRx in the US and some additional markets
- Under this collaboration, Akcea received an up front payment of $250M and was eligible to receive $1.3B in development, regulatory, and commercial milestones along with mid-teens to low 20% tiered royalties. In Jan 2022, following the data highlights from the P-II clinical evaluation of AKCEA-ANGPTL3-LRx, Pfizer stepped back from the collaboration agreement

GSK Terminated its Research Partnership with Lyell Immunopharma
- In May 2019, GSK signed a 5-year research collaboration with Lyell Immunopharma to develop cell therapies including GSK3377794 (targeting NY-ESO-1 antigen) by utilizing Lyell's technologies for the treatment of solid tumors. Lyell also received an option to license 7 additional targets. As per the terms of the agreement, Lyell received $45M up front and up to $200M for validating its T-cell modulating technology in a clinical setting
- Additionally, Lyell was eligible for up to $1.3B in development and commercial milestones out of which $400M was marked per selected target present within GSK’s pipeline and $900M per selected target not present within GSK’s pipeline
- Lyell was also eligible for single-digit to low-teens royalties. Pertaining to the GSK agreement, Lyell also entered into a Stock Purchase Agreement with GSK in terms of which Lyell sold and issued 30,253,189 shares of Series AA convertible preferred stock at a price of $6.78 per share. Later in Oct 2022, GSK terminated this cell therapy deal with Lyell

Otsuka Pharmaceutical Terminated its Collaboration and License Agreement with Akebia Therapeutics
- In May 2019, GSK signed a 5-year research collaboration with Lyell Immunopharma to develop cell therapies including GSK3377794 (targeting NY-ESO-1 antigen) by utilizing Lyell's technologies for the treatment of solid tumors. Lyell also received an option to license 7 additional targets. As per the terms of the agreement, Lyell received $45M up front and up to $200M for validating its T-cell modulating technology in a clinical setting
- Additionally, Lyell was eligible for up to $1.3B in development and commercial milestones out of which $400M was marked per selected target present within GSK’s pipeline and $900M per selected target does not present within GSK’s pipeline. Lyell was also eligible for single-digit to low-teens royalties. Pertaining to the GSK agreement, Lyell also entered into a Stock Purchase Agreement with GSK in terms of which Lyell sold and issued 30,253,189 shares of Series AA convertible preferred stock at a price of $6.78 per share
- In Dec 2021, Lyell and GSK amended the terms of their agreement based on which Lyell took the responsibility to manufacture the NYESO-1 + Epi-R TCR cell therapy product for an initial planned P-I clinical trial (Epi-R Trial) at its manufacturing facility in Bothell, Washington and will be responsible for submitting the IND to the US FDA for the product. On the other hand, GSK was responsible to conduct the Epi-R Trial under its First Time in Humans Master Protocol for NY-ESO-1 (FTIH Protocol) based on a mutually agreed clinical plan and was also responsible to file its updated FTIH protocol and for regulatory interactions with the US FDA. Later in Oct 2022, GSK terminated this cell therapy deal with Lyell

BMS Terminated its Development and Commercialization Deal with uniQure
- In Apr 2015, uniQure granted BMS the exclusive global rights to develop and commercialize uniQure’s gene therapy technology platform for specific collaboration targets including S100A1 for the treatment of CVS diseases. As per the terms of the agreement, uniQure received an up front payment of $50M, $15M for the selection of 3 collaborative targets including S100A1, and ~$75.5M in 2 equity investments
- uniQure was also eligible to receive up to $254M for S100A1 and up to ~$217M each for other gene therapy products in research, development, and regulatory milestone and undisclosed sales-based milestones plus royalties. Later in Jun 2015, BMS purchased 1.1M ordinary shares at $33.84 per share for a total of ~$37.6M. In Feb 2019, BMS requested a one-year extension of the initial research term which was declined by uniQure in Apr 2019
- An amendment to the agreement took place in Dec 2020 through which BMS was able to replace up to 2 of the 4 active collaboration targets with 2 new targets for which uniQure would receive $217M for each of the four targets. The amendment also terminated two warrants to increase BMS’ ownership to up to 19.9% through the purchase of a specific number of ordinary shares. Additionally, if BMS delivers a target cessation notice for all 4 targets then uniQure would have to pay BMS a one-time, non-refundable, non-creditable cash payment of $70M. In Nov 2022, BMS reported the termination of its collaboration with uniQure
Related Posts: Top 20 Biopharma Deal Terminations of 2021 Based on Total Deal Value
Tags

Shivani was a content writer at PharmaShots. She has a keen interest in recent innovations in the life sciences industry. She was covering news related to Product approvals, clinical trial results, and updates. We can be contacted at connect@pharmashots.com.