
BioMarin Announces Positive Phase 3 Gene Therapy Trial Results in Adults with Severe Hemophilia A; Study Met All Primary and Secondary Efficacy Endpoints in One-Year Data Set

- Significantly Reduced Mean Annualized Bleeding Rate by 84% (p-value <0.0001) Demonstrating Superiority to Factor VIII Prophylaxis, and Reduced Mean Annualized Factor VIII Infusion Rate by 99% (p-value <0.0001)
- Mean Factor VIII Expression 42.9 IU/dL at One Year in Full Study Population
- In Subset Dosed More than Two Years Ago, Slower Rate of Decline in Factor VIII Expression Was Observed Compared to Prior Study: Mean ABR in This Population Was 0.9 Over These Two-Plus Years
- BioMarin Plans to Meet with EMA to Discuss Submission of One-Year Data and FDA to Review Two-Year Data Request
- Call and Webinar to be Held Today, Sunday, January 10, 2021 at 7:15 PM Eastern
SAN RAFAEL, Calif.,?Jan. 10, 2021?/PRNewswire/ --?BioMarin Pharmaceutical Inc. (NASDAQ:?BMRN) today announced positive topline results from its ongoing global Phase 3 GENEr8-1 study of valoctocogene roxaparvovec, an investigational gene therapy for the treatment of adults with severe hemophilia A.? This is the largest global Phase 3 study to date for any gene therapy in any indication, with 134 participants. ?All participants in the study received a single dose of valoctocogene roxaparvovec and completed a year or more of follow-up.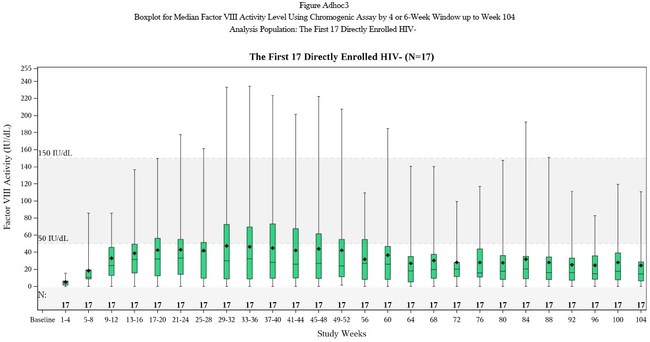
|
Phase 3 Rollover Population* On Factor VIII prophylaxis, before N=112 |
Phase 3 Rollover Population* After valoctocogene roxaparvovec N=112 |
|
|
Mean (SD) Median (IQR) |
Mean (SD) Median (IQR) |
|
|
Annualized Bleeding Rate (bleeding episodes per |
4.8 (6.5) 2.8 (0.0, 7.6) |
0.8 (3.0) 0.0 (0.0, 0.0) |
|
Annualized FVIII??Infusion year) |
135.9 (52.0) 128.6 (104.1, 159.9) |
2.0 (6.4) 0.0 (0.0, 0.9) |
|
*See study descriptions for patient population information. |
|
Median Factor VIII Activity, IU/dL |
Phase 3 Rollover Population (N=112) Mean (SD) Median |
Phase 3 mITT Subset Population (N=17*) Mean (SD) Median |
Phase 1/2 6e13 vg/kg Cohort (N=7) Mean (SD) Median |
Phase 1/2 4e13 vg/kg Cohort (N=6) Mean (SD) Median |
|
Week 26 |
55.1 (57.4) 38.6 |
43.9 (42.1) 33.8 |
71.0 (41.6) 61.2 |
18.0 (8.7) 18.0 |
|
Week 52 |
43.6 (45.3) 24.2 |
42.2 (50.9) 23.9 |
63.6 (36.5) 60.3 |
21.1 (12.3) 23.8 |
|
Week 76 |
27.9 (30.6) 15.8 |
53.9 (31.2) 50.2 |
20.6 (15.4) 21.3 |
|
|
Week 104 |
24.4 (29.2) 14.7 |
36.4 (26.3) 26.2 |
12.3 (8.2) 11.6 |
|
*Includes only HIV-negative subjects dosed 2 or more years prior to Nov 2020 data cut date. One participant was lost to follow-up at 66.1 weeks and was henceforth imputed to have a Factor VIII activity of 0 IU/dL through 104 weeks. |



