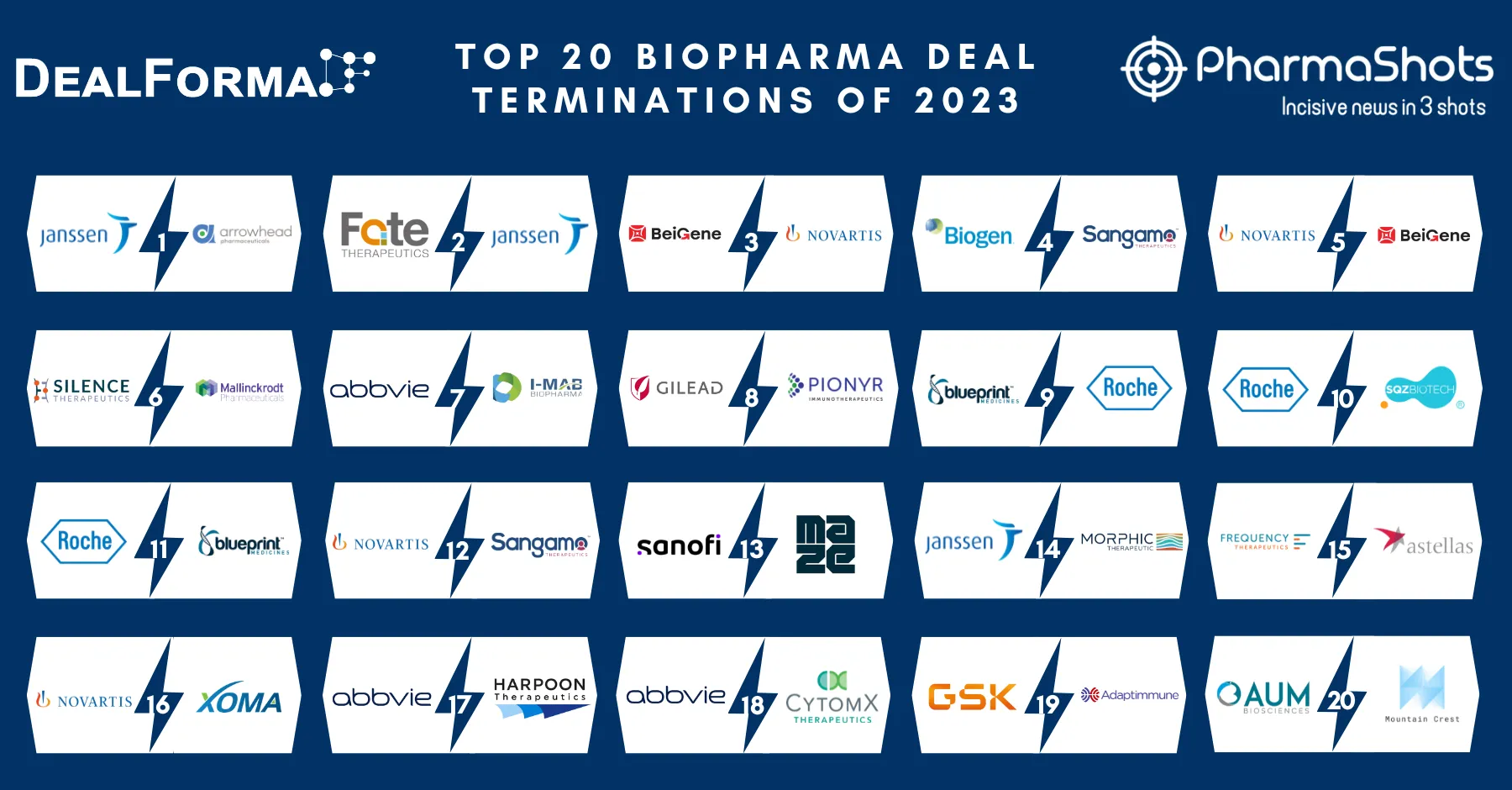
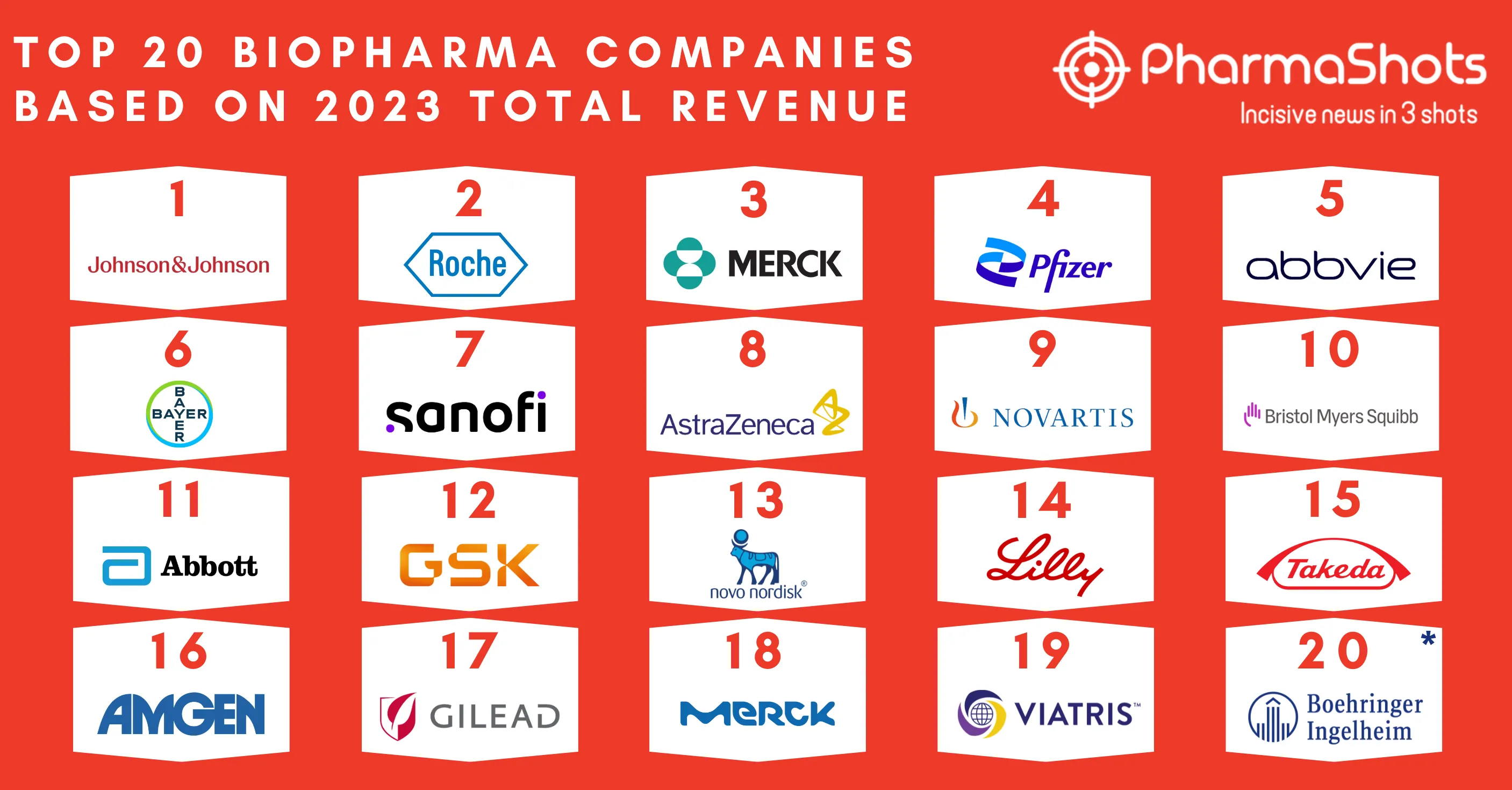
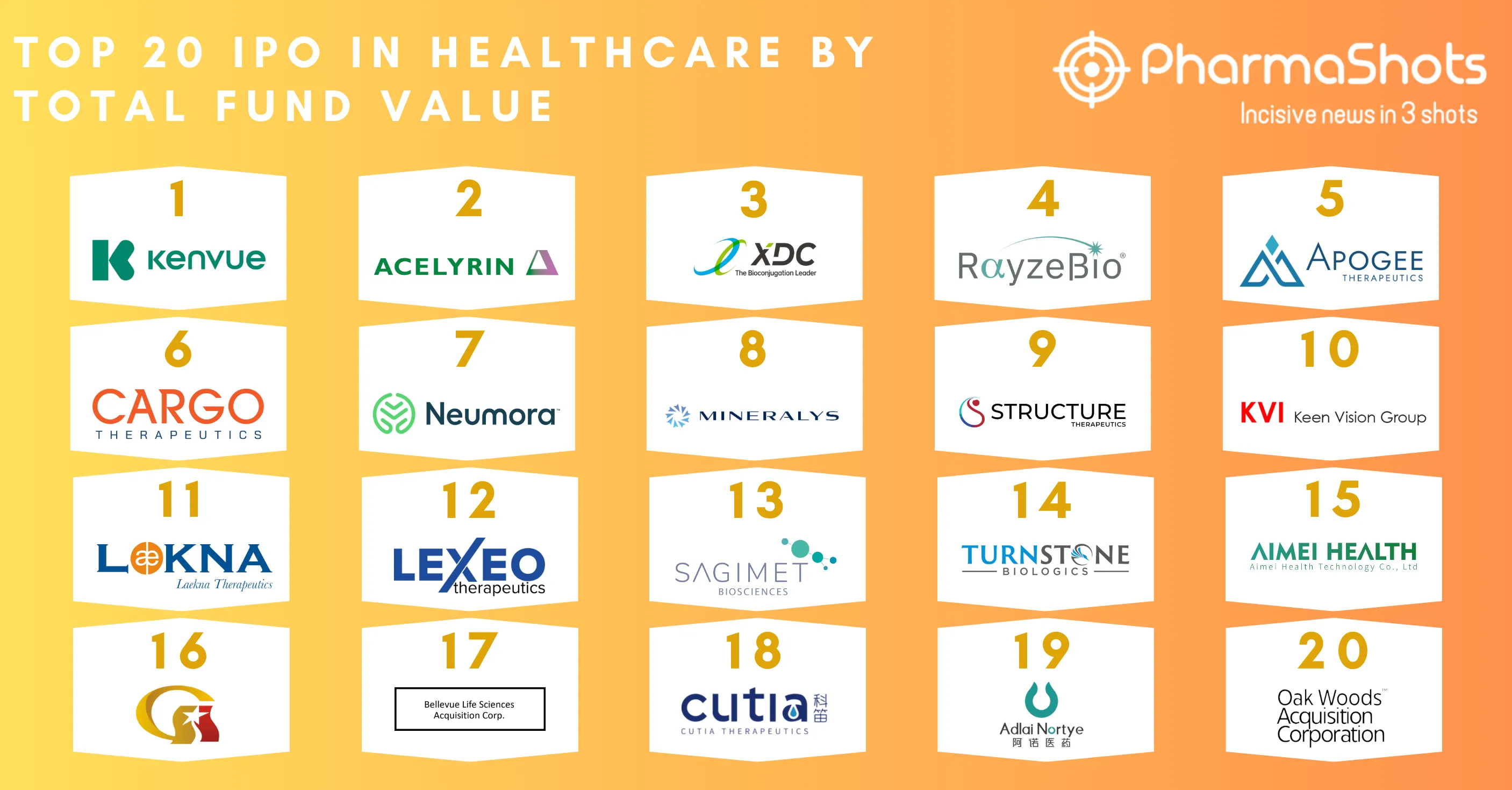
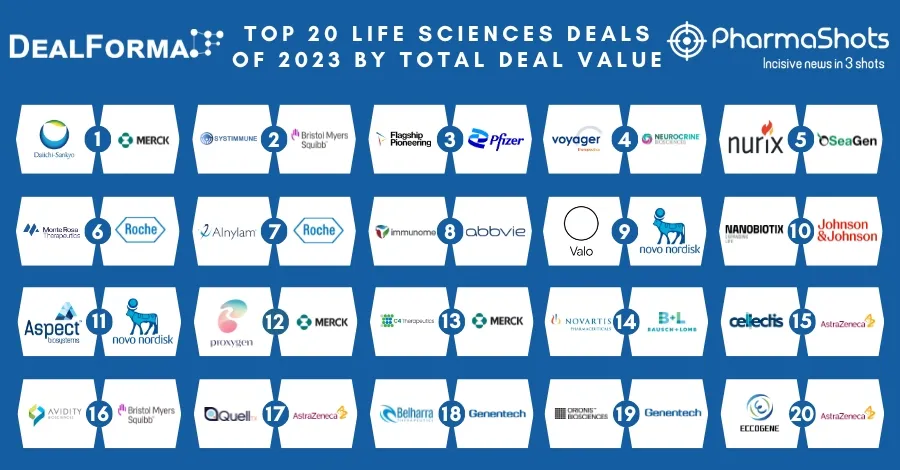
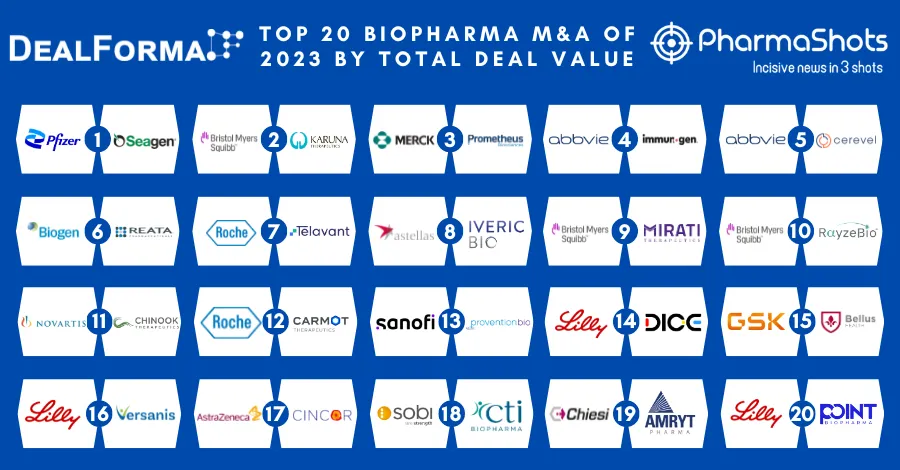
Top 20 Prescription Drugs Based on 2019 Revenue
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs or medicines are being produced for a wide range of medical sectors. It includes the lifesaving drugs or the major therapy area including immunology- cardiology- and neurology but are they not limited to only these indications and are rapidly increasing with the increasing medical needs. The drugs are developed targeting with the motive to cure- vaccinate- and alleviate the symptoms.
We have compiled a list of global top 20 drugs blockbuster prescription drugs based on their sales for last year i.e. 2019. The top position was maintained by AbbVie's blockbuster drug Humira with $19.16B another drug grasped the second position headed by Merck's Keytruda with $11.08B following the third was occupied by BMS's Revlimid with $9.37B while ended at the low end by Gilead's Truvada.
If you have any questions or see something we might have missed- Please reach out to Senior Editor- Shiwani Sharma by email.
Product - Truvada
First Approved - US (Aug 02- 2004)- EU (Feb 20- 2005)
Indications Approved - HIV-1
Company - Gilead Sciences
Total Revenue - $2.81B
Truvada is a combination of tenofovir disiproxil fumarate (tenofovir DF) and emtricitabine used for HIV treatment and pre-exposure prophylaxis for PrEP (pre-exposure prophylaxis) that can help reduce the risk of getting HIV-1 through sex. On July 23- 2019- Gilead presented new findings on the profile of Descoy for potential use as HIV Pre-exposure Prophylaxis compared with Truvada. Descovy and Truvada are only 2 FDA-approved pills for PrEP.
Product - Neulasta
First Approved - US (Jan 31- 2002)- EU (Aug 22- 2002)
Indications Approved - Febrile Neutropenia
Company - Amgen
Total Revenue - $3.22B
Neulasta is a PEGylated form of the recombinant human granulocyte colony-stimulating factor analogue filgrastim. Neulasta has been steadily losing market share due to the launch of multiple biosimilars and the utilization of Neulasta's OnPro on-body injector (the dominant player at > 55%) has been relatively steady.
Product - Victoza Company - Novo Nordisk
First Approved - US (Jan 25- 2010)- EU (Jun 30- 2009) Total Revenue - $3.29B
Indications Approved - Glycemic control in type 2 diabetes mellitus patients- Reduce adverse cardiovascular events in adults with type 2 diabetes mellitus
Victoza or liraglutide belongs to a class of drugs called glucagon-like peptide-1 agonists (GLP-1). Recently Novo Nordisk's patent on Victoza is been reviewed as Mylan has claimed that its invalid because it covers an obvious invention. Mylan has challenged the Victoza patent with an aim to launch the low-cost copy of the drug.
Product - Lyrica Company - Pfizer
First Approved - US (Dec 30- 2004)- EU (Jul 05- 2004) Total Revenue - $3.32 B
Indications Approved - Neuropathic pain associated with diabetic peripheral neuropathy- Postherpetic neuralgia
Lyrica is an anticonvulsant or an anti-seizure drug that can treat a range of conditions- including epilepsy- fibromyalgia- and nerve pain. The recent update on May 28- 2019- Pfizer announced that its epilepsy drug Lyrica (pregabalin)- failed to meet the primary endpoint in P-III study- assessing it as adjunctive therapy in epilepsy patients (aged 5 to 65 years) with primary generalized tonic-clonic ('PGTC') seizures.
Product - Imbruvica Company - Johnson & Johnson
First Approved - US (Nov 13- 2013)- EU (Nov 21- 2014) Total Revenue - $3.41B
Indications Approved - Mantle Cell Lymphoma- Chronic Lymphocytic Leukemia
Imbruvica is an oral therapy which inhibits Bruton's tyrosine kinase (BTK) and has received its 10 FDA approval. In Apr 2020- US FDA approved Imbruvica (ibrutinib) plus Rituximab for the treatment of patients with Chronic Lymphocytic Leukemia (CLL)
Product - Genvoya Company - Gilead Sciences
First Approved - US (Nov 05- 2015)- EU (Nov 19- 2015) Total Revenue - $3.93 B
Indications Approved - HIV-1
Genvoya is a combination of elvitegravir- cobicistat- emtricitabine- and tenofovir alafenamide targeting HIV. In Aug 2018- The CNDA approved Gilead's Genvoya (elvitegravir 150 mg/cobicistat 150 mg/emtricitabine 200 mg/tenofovir alafenamide 10 mg or E/C/F/TAF) for treating HIV Infection.
Product - Remicade Company - Johnson & Johnson
First Approved - US (Aug 24- 1998)- EU (Aug 13- 1999) Total Revenue - $4.38B
Indications Approved - Crohn's Disease- Rheumatoid Arthritis- Ankylosing Spondylitis- Plaque Psoriasis- Psoriatic Arthritis- Pediatric Ulcerative Colitis- Pediatric Crohn's Disease
Remicade or infliximab is a tumour necrosis factor (TNF-alpha or TNF-a) blocker and a chimeric monoclonal IgG1 antibody. In June 2019- Janssen's Remicade was issued a Civil Investigative Demand to Johnson & Johnson by FTC for investigating whether its contracting practices violate federal antitrust.
Product - Ibrance Company - Pfizer
First Approved - US (Feb 03- 2015)- EU (Nov 09- 2016) Total Revenue - $4.96B
Indications Approved - HER2 Negative Advanced Breast Cancer
Ibrance PO is CDKs 4 and 6 inhibitors indicated for HR+- HER2- advance or mBC and has been prescribed up xto 160-000 patients with approval in 85 countries worldwide. On May 29- 2020- the Data Monitoring Committee (DMC) of the collaborative P-III early breast cancer PALbociclib CoLlaborative Adjuvant Study (PALLAS) determined that the trial failed in the primary endpoint of invasive disease-free survival (iDFS). Eli Lilly's Verzenio has been chasing Ibrance in the metastatic setting since it hit the market and it now its ahead of Ibrance which recently took a big blow.

Product - Enbrel Company - Amgen
First Approved - US (Nov 02- 1998)- EU (Feb 02- 2000) Total Revenue - $5.22B
Indications Approved - Rheumatoid Arthritis- Polyarticular-Course Juvenile Rheumatoid Arthritis- Psoriatic Arthritis- Ankylosing Spondylitis- Plaque Psoriasis
Enbrel is a TNF inhibitor drug that treats autoimmune diseases by interfering with tumour necrosis factor by acting as a TNF inhibitor. Enbrel was the drug of choice for multiple autoimmune indications. On Jul 01- 2020- the US Court of Appeals for the Federal Circuit held in Amgen's favour on the validity of 2 patents that describe and claim Enbrel's methods.
Product - Prevnar 13 Company - Pfizer
First Approved - US (Feb 24- 2010)- EU (Dec 12- 2009) Total Revenue - $5.84B
Indications Approved - S. Pneumoniae Infection
Prevnar 13 or Pneumococcal 13-valent Conjugate Vaccine [Diphtheria CRM197 Protein is a vaccine approved for adults 18 years of age and older for the prevention of pneumococcal pneumonia and invasive disease caused by 13 Streptococcus pneumonia strains. In June 2020- Pfizer started four P-III clinical trials for investigational vaccines including Prevnar 13.

Product - Herceptin Company - Roche
First Approved - US (Sep 25- 1998)- EU (Aug 28- 2000) Total Revenue - $6.23 B
Indications Approved - Metastatic Breast Cancer- Metastatic Gastric or Gastroesophageal Junction Adenocarcinoma
Herceptin or trastuzumab is a mAb that binds to HER2 receptors on the surface of HER2-positive tumor cells- blocking them from receiving growth signals and flagging them for destruction by the immune system. It is on the WHO's List of Essential Medicines- the safest and most effective medicines needed in a health system. On 29 June 2020- Roche got the approval of Phesgo- a fixed-dose combination of Perjeta (pertuzumab) and Herceptin with hyaluronidase- administered by SC injection in combination with IV chemotherapy- for the treatment of early and metastatic HER2-positive breast cancer. This is the first time that Roche has combined two mAbs that can be administered by a single SC injection.
Product - Stelara Company - Johnson & Johnson
First Approved - US (Sep 25- 2009)- EU (Jan 15- 2009) Total Revenue - $6.36B
Indications Approved - Plaque Psoriasis- Psoriatic Arthritis- Crohn's Disease- Ulcerative Colitis
Stelara or ustekinumab is a mAb with a novel mechanism of action that targets the p40 subunit of cytokines interleukin-12 (IL-12) and interleukin-23 (IL-23). In Apr 2020- Johnson & Johnson received NICE's positive Final Appraisal Document (FAD) recommending Stelara (ustekinumab) for the treatment of ulcerative colitis.

Product - Rituxan Company - Roche
First Approved - US (Nov 26- 1997)- EU (Jun 02- 1998) Total Revenue - $6.69B
Indications Approved - Non-Hodgkin's lymphoma- Chronic Lymphocytic Leukemia- Rheumatoid Arthritis- Granulomatosis with Polyangiitis- Microscopic Polyangiitis- Pemphigus Vulgaris
Rituxan or rituximab is a mAb used to target cancer cells with CD20 markers in patients. In Sep 2019- The US FDA approved Roche's Rituxan (rituximab) in combination with glucocorticoids for treating granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) disorders.
Product - Opdivo Company - Bristol-Myers Squibb
First Approved - US (Dec 22- 2014)- EU (Jun 19- 2015) Total Revenue - $7.20B
Indications Approved - Metastatic Melanoma- Non-Small Cell Lung Cancer-Melanoma- Small Cell Lung Cancer- Renal Cell Carcinoma- Hodgkin Lymphoma- Squamous Cell Carcinoma of the Head and Neck- Urothelial Carcinoma- Mismatch Repair Deficient Metastatic Colorectal Cancer- Hepatocellular Carcinoma- Esophageal Squamous Cell Carcinoma
Opdivo or nivolumab is a PD-1 immune checkpoint inhibitor targeted for cancer cells and is approved in 65 countries including the US- EU- Japan & China. In Jun 2020- the US FDA granted approval for Opdivo (nivolumab) following the P-III ATTRACTION-3 study results evaluate it in comparison with taxane CT (docetaxel/paclitaxel) to treat patients with unresectable advanced- recurrent or ESCC- refractory/ intolerant to at least one prior fluoropyrimidine & platinum-based CT.

Product - Avastin Company - Roche
First Approved - US (Feb 26- 2004)- EU (Jan 12- 2005) Total Revenue - $7.30B
Indications Approved - Metastatic Carcinoma- Metastatic Colorectal Cancer- Non-Small Cell Lung Cancer- Glioblastoma- Metastatic Renal Cell Carcinoma- Metastatic Cervical Cancer- Primary Peritoneal Cancer- Hepatocellular Carcinoma
Avastin or bevacizumab is a tumour-starving (anti-angiogenic) therapy targeted for preventing tumour growth. In Jun 2020- Roche received the US FDA's approval for a combination of Avastin (bevacizumab) + Tecentriq (atezolizumab) to treat patients with Unresectable Or Metastatic Hepatocellular Carcinoma (HCC) who have not received prior systemic therapy- which lead to a novel immunotherapy approval for the indication. The approval followed the P-III IMbrave150 study results assessing the combination.
Product – Eylea Company - Regeneron Pharmaceuticals
First Approved - US (Nov 18, 2011), EU (Nov 21, 2012) Total Revenue - $7.54B
Indications Approved - Age-Related Macular Degeneration, Macular Edema, Diabetic Macular Edema, Diabetic Retinopathy
Eylea or aflibercept is one form of anti-VEGF therapy administered by injection into the eye. Recently, Bayer launched Eylea (aflibercept) pre-filled syringe in all 27 states of the EU including the UK, Iceland, Norway, and Liechtenstein in Apr 2020. In May 2019, Regeneron’s Eylea injection received the US FDA’s approval to treat all stages of Diabetic Retinopathy (DR) further reducing the risk of blindness.
Related Post: Top COVID-19 Deals (Part III): Biopharma and Govt. Institutions
Tags

This content piece was prepared by our former Senior Editor. She had expertise in life science research and was an avid reader. For any query reach out to us at connect@pharmashots.com