Insights+: The US FDA New Drug Approvals in July 2023
Shots:
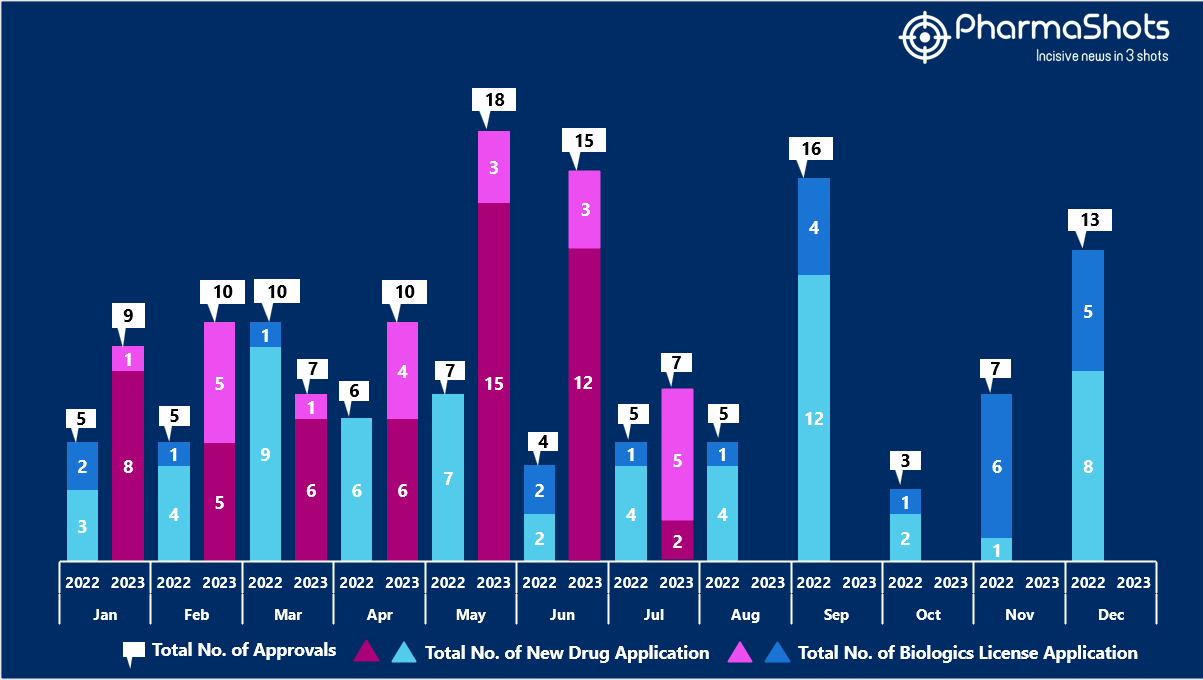
- The US FDA approved 5 NDAs and 2 BLA in July 2023, leading to treatments for patients and advances in the healthcare industry. The CDER and CBER approved 76 novel products in 2023
- In July 2023, the major highlights drugs were Beyfortus (nirsevimab) approval for the prevention of RSV lower respiratory tract disease in infants, Vanflyta for newly diagnosed FLT3-ITD positive acute myeloid leukemia
- PharmaShots has compiled a list of a total of 7 new drugs approved by the US FDA in July 2023
Beyfortus
Active ingredient: nirsevimab Approved: July 19, 2023
Company: AstraZeneca and Sanofi Disease: RSV Lower Respiratory Tract Disease
- The US FDA has approved Beyfortus for RSV LRTD in newborns & infants born during or entering their first RSV season, and for children ~24mos. who remain vulnerable to sev. RSV disease through the 2nd RSV season. The therapy is expected to be available in the 2023-2024 RSV season
- The approval was based on the extensive clinical development program incl. 3 late-stage trials evaluating Beyfortus. The results showed that a single dose of Beyfortus demonstrated consistent efficacy against RSV LRTD through 5mos. across all clinical EPs, was well tolerated with a favorable safety profile consistent & overall rates of AEs were comparable b/w Beyfortus & PBO
- The therapy was approved in the EU, Great Britain & Canada while regulatory applications are under review in China, Japan & multiple other countries
Vanflyta
Active ingredient: quizartinib Approved: July 21, 2023
Company: Daiichi Sankyo Disease: Acute Myeloid Leukemia
- The US FDA has approved Vanflyta in combination with standard cytarabine and anthracycline induction and cytarabine consolidation & as maintenance monotx., following consolidation CT for adult patients with newly diagnosed AML i.e., FLT3-ITD+
- The approval was based on the P-III trial (QuANTUM-First) results published in The Lancet evaluating Vanflyta + standard induction and consolidation therapy incl. HSCT & as maintenance monotx. in a ratio (1:1) in 539 patients aged 18-75yrs. at 193 study sites across Asia, the EU, North & South America, and Oceania
- The results showed a 22% reduction in risk of death, CR rates were similar b/w both arms, and the median duration of CR (38.6 vs 12.4mos.) in PBO + standard CT alone. The therapy is expected to be available by prescription in the US in the coming weeks
NexGard PLUS
Active ingredient: afoxolaner, moxidectin, and pyrantel Approved: July 21, 2023
Company: Boehringer Ingelheim Disease: Internal and External Parasites
- The US FDA has approved afoxolaner, moxidectin & pyrantel chewable tablets (NexGard PLUS), a new one-and-done monthly combination product for canine protection from fleas, ticks, heartworm disease, roundworms & hookworms
- Multiple clinical studies showed that NexGard PLUS was safe & effective, ≥99.8% effective 24hrs. in killing adult fleas fast after weekly infestations for a full month after treatment, 100% effective in preventing heartworm disease in dogs, safe for puppies as young as 8wks. & in P-gp-deficient (MDR1-mutant) avermectin-sensitive dogs
- Boehringer Ingelheim’s beef-flavored afoxolaner, moxidectin & pyrantel chewable tablets are expected to be available for veterinary clinics on July 2023
Ycanth
Active ingredient: cantharidin Approved: July 24, 2023
Company: Verrica Pharmaceuticals Disease: Molluscum Contagiosum
- The US FDA has approved Ycanth (cantharidin) topical solution for adult & pediatric patients aged ≥2yrs. with molluscum contagiosum. The approval was based on 2 P-III trials (CAMP-1 & 2) that evaluated VP-102 (Ycanth) vs PBO in patients aged ≥2yrs.
- Both trial results showed that patients treated with VP-102 met its 1EPs of complete clearance of all treatable molluscum lesions, 46% vs 18% achieved complete clearance of molluscum lesions in the (CAMP-1) trial while 54% vs 13% in the (CAMP-2) trial
- In an additional post-hoc analysis, complete clearance of all lesions was higher in the VP-102 group vs vehicle across all body regions. Ycanth, a drug-device combination product administered by a healthcare professional is expected to be available in Sept 2023
Xdemvy
Active ingredient: lotilaner Approved: July 27, 2023
Company: Tarsus Pharmaceuticals Disease: Demodex Blepharitis
- The US FDA has approved Xdemvy, the first and only US FDA-approved treatment to directly target Demodex mites for the treatment of Demodex blepharitis. Xdemvy is expected to be available by prescription at the end of Aug 2023
- The approval was based on two studies (Saturn-1 & 2) evaluating Xdemvy vs vehicle in a ratio (1:1) in 833 patients which showed an improvement in eyelids (reduction of collarettes, the pathognomonic sign of the disease, to no more than 2 collarettes per upper lid) in each study by Day 43 with some patients showed improvement as early as 2wks.
- The EPs of mite eradication & erythema cure showed an improvement at Day 43 across both studies, and were safe & well tolerated while other ocular adverse reactions were reported in ≤2% of patients
6. BioMarin’s Roctavian Receives the US FDA’s Approval for the Treatment of Severe Hemophilia A
Roctavian
Active ingredient: valoctocogene roxaparvovec Approved: July 30, 2023
Company: BioMarin Disease: Hemophilia A
- The approval was based on the P-III (GENEr8-1) study evaluating Roctavian in patients (n=134) with severe hemophilia A. For 112 patients, baseline ABR data were collected during 6mos. on FVIII prophylaxis before receiving Roctavian & for 22 patients, baseline ABR was collected retrospectively
- The results showed that in 112 patients, a mean ABR reduction of 52% was reported after receiving Roctavian by the end of follow-up (2.6 bleeds/year) vs baseline ABR while receiving routine FVIII prophylaxis (5.4 bleeds/year) & rate of spontaneous bleeds & joint bleeds was also reduced with Roctavian (0.5 & 0.6 vs 2.3 &3.1 bleeds/year)
- BioMarin expects to continue to monitor the long-term effects of the treatment with a follow-up of 15yrs.
Jemperli
Active ingredient: dostarlimab Approved: July 31, 2023
Company: GSK Disease: Endometrial Cancer
- The approval of Jemperli (dostarlimab-gxly) + carboplatin + paclitaxel, followed by Jemperli as monotx. for the treatment of adult patients with dMMR primary advanced or recurrent endometrial cancer
- This approval is based on the interim analysis results from Part 1 of the P-III study (RUBY), which demonstrated a median duration of follow-up of ≥25mos. A significant and clinically meaningful benefit in PFS and a 71% reduction in the risk of disease progression or death. The trial continues to evaluate OS
- Consistent safety and tolerability were seen over the monotherapies. The most common TEAEs were ≥20%. Also, the data were presented at ESMO and SGO 2023 and published in The New England Journal of Medicine
Related Post: Insights+: The US FDA New Drug Approvals in June 2023





